Pharmacovigilance (PV)
What is Pharmacovigilance?
Pharmacovigilance (PV) is the science and set of activities related to the detection, assessment, understanding, and prevention of adverse effects or any other drug-related problems.
Its primary goal is to improve patient safety and ensure that the benefits of a medicinal product outweigh its risks throughout its lifecycle — from clinical development through post-marketing.
Pharmacovigilance is both a regulatory requirement and a public health priority, with systems and processes in place globally to monitor the safety of medicinal products in real time.
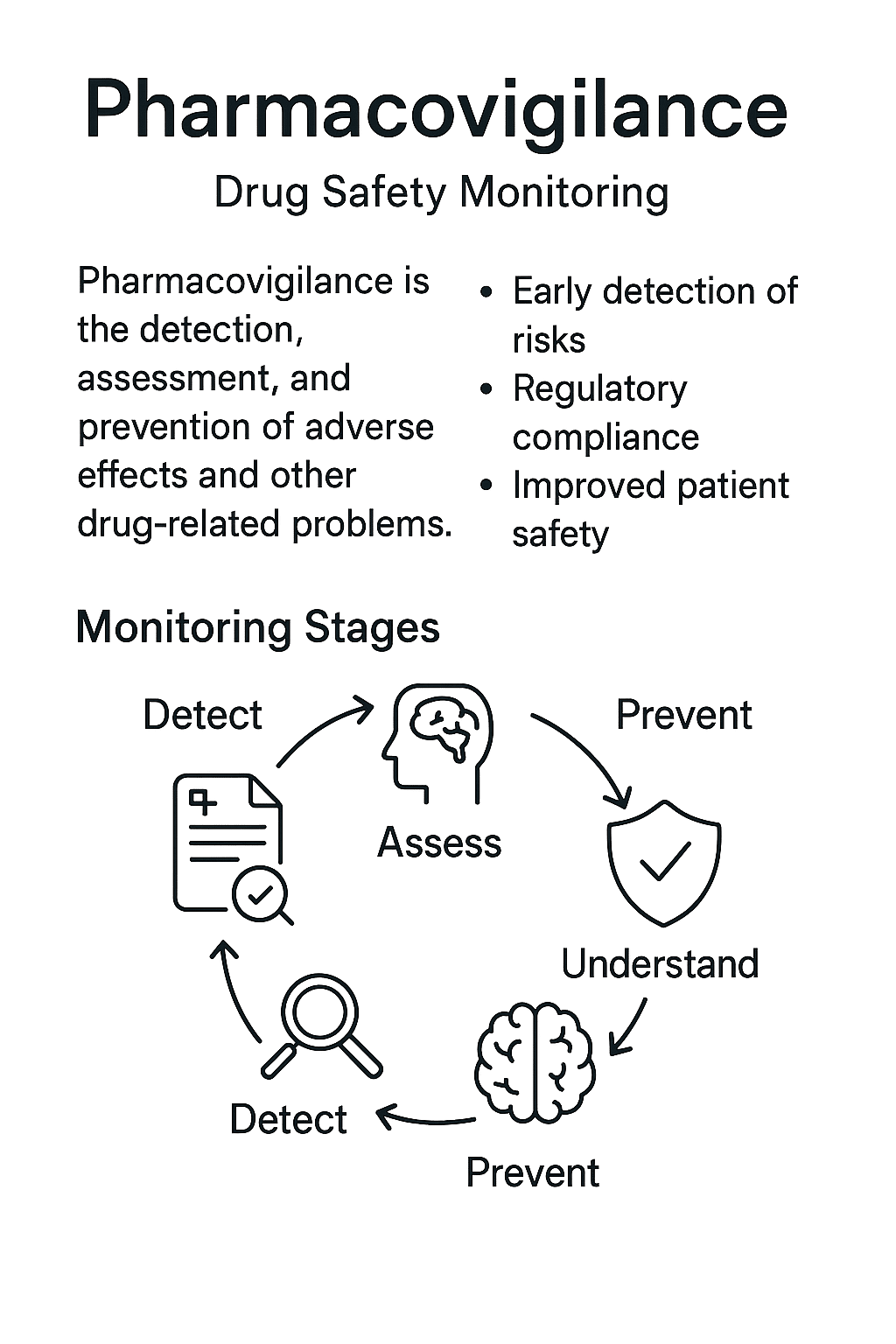
Why it Matters in Pharma Today
- Regulatory compliance — Required by global health authorities for all marketed products, with defined processes for reporting and evaluation.
- Patient safety — Enables rapid identification and mitigation of emerging safety risks.
- Evidence-based decisions — Provides data to inform labeling updates, risk minimisation measures, and benefit–risk assessments.
- Global harmonisation — Aligns safety monitoring practices through international standards (ICH E2 series, WHO Uppsala Monitoring Centre).
- Data integration — PV data feeds directly into labeling governance (CCDS, SmPC, USPI, ePI) in structured content workflows.
Global Landscape
| Region/Body | Governing Entity | Adoption Status / Timeline |
| Global | WHO Uppsala Monitoring Centre (UMC) | Maintains VigiBase, the global database of individual case safety reports (ICSRs). |
| EU | EMA & National Competent Authorities | Governed by Good Pharmacovigilance Practices (GVP) modules. |
| US | FDA (CDER & CBER) | Adverse Event Reporting System (FAERS) for marketed drugs and biologics. |
| Japan | PMDA | Maintains national pharmacovigilance database; aligns with ICH standards. |
How Pharmacovigilance Works in a Component-Based Authoring Model
Detect — Identify potential safety issues through adverse event reports, literature monitoring, signal detection tools, and ongoing clinical trial surveillance.
Assess — Evaluate the causality, frequency, and severity of identified risks.
Understand — Integrate safety data with clinical and product information to assess the overall benefit–risk profile.
Prevent — Implement risk minimisation actions, including labeling changes, safety communications, or product withdrawal if necessary.
Propagate changes — Update CCDS, SmPC, USPI, ePI, and other documents using structured content to ensure consistency across all outputs.
Example: PV in Practice
- New drug–drug interaction detected → PV team updates CCDS → triggers updates to SmPC Section 4.5 and USPI Section 7 → regulatory submissions made to all affected markets.
- Unexpected adverse event in post-market use → PV signal assessment recommends boxed warning → labeling updates implemented globally.