The Importance of Structured Content Authoring for ePI Compliance
What is ePI?
Electronic Product Information (ePI) is the digital version of essential medicine information, including patient leaflets and prescribing details. Unlike traditional PDFs, ePI is structured, machine-readable, and designed for seamless integration into healthcare systems. It aligns with globally accepted regulatory frameworks from leading authorities and organizations, such as:
- FDA SPL (Structured Product Labeling)
- EMA ePI (European Medicines Agency’s ePI standard)
- FHIR (Fast Healthcare Interoperability Resources)
- National agency frameworks, such as the Jordan FDA (JFDA) and Health Canada initiatives
By transitioning to structured ePI formats like XML and JSON, regulatory bodies and pharmaceutical companies can ensure greater accessibility, accuracy, and compliance with evolving digital health standards while avoiding the cost of manual solutions.
Global Adoption of ePI and Similar Regulations
Many countries are moving toward ePI adoption to enhance regulatory efficiency and improve patient access to accurate product information.
- European Union: The European Medicines Agency (EMA) is leading the transition to ePI, with several countries—including Denmark (DKMA), the Netherlands (MEB), Spain (AEMPS), and Sweden (MPA—participating in pilot programs.
- Belgium (FAMHP), Luxembourg (DPM), Germany (BfArM), Spain (AEMPS), and the Baltic States: These nations have ongoing ePI initiatives and pilot projects exploring the shift from paper-based to electronic product information.
- Jordan: The Jordan Food and Drug Administration (JFDA) mandated mid-January 2025 emerging ePI standards must be followed, setting an example for other regions.
- Canada: Health Canada has introduced electronic drug information initiatives, such as guidance for electronic media-based prescription drug labeling and the Electronic Canadian Drug Facts Table (eCDFT) for non-prescription drugs. These efforts align with the global trend toward digital product information and improved patient accessibility.
- United States of America: U.S. Food & Drug (FDA) has been on record since 2005 transitioning to ePI SPL for the United States Prescribing Information (USPI) to allow the exchange of information in a way that cannot be accomplished with PDF.
With different regulatory bodies taking steps toward structured electronic labeling, the ability to create and manage adaptable content is becoming increasingly important.
How Structured Content Authoring Facilitates and Accelerates ePI Compliance
Structured content authoring separates content from its presentation, ensuring flexibility, consistency, and compliance with industry regulations. This approach is essential for ePI management, providing key advantages such as:
Ensuring Consistency & Compliance
- Maintains uniformity across regulatory submissions, reducing errors.
- Adapts seamlessly to new compliance standards such as FHIR-based ePI.
Improving Content Reusability & Adaptability
- Enables repurposing of product information across multiple platforms, including electronic health records, mobile apps, and regulatory portals.
- Ensures fidelity of content through what has historically been a transformative process.
- Facilitates easy updates without disrupting other content components.
Enhancing Machine-Readability & Automation
- Supports advanced metadata tagging, meeting critical Identification of Medicinal Product (IDMP) and other data rich regulations
- Highly indexable, improving searchability and retrieval.
- Ensures seamless system-to-system communication between pharmaceutical companies and regulatory agencies.
Simplifying Regulatory Submissions & Feedback Loops
- Structured ePI content integrates effortlessly into regulatory reviews.
- Streamlines secure data exchanges while maintaining content integrity.
Supporting Global Compliance & Interoperability
- Aligns with international ePI frameworks for seamless regulatory adoption.
- Ensures product information is accessible and structured for healthcare professionals, patients, and regulators worldwide.
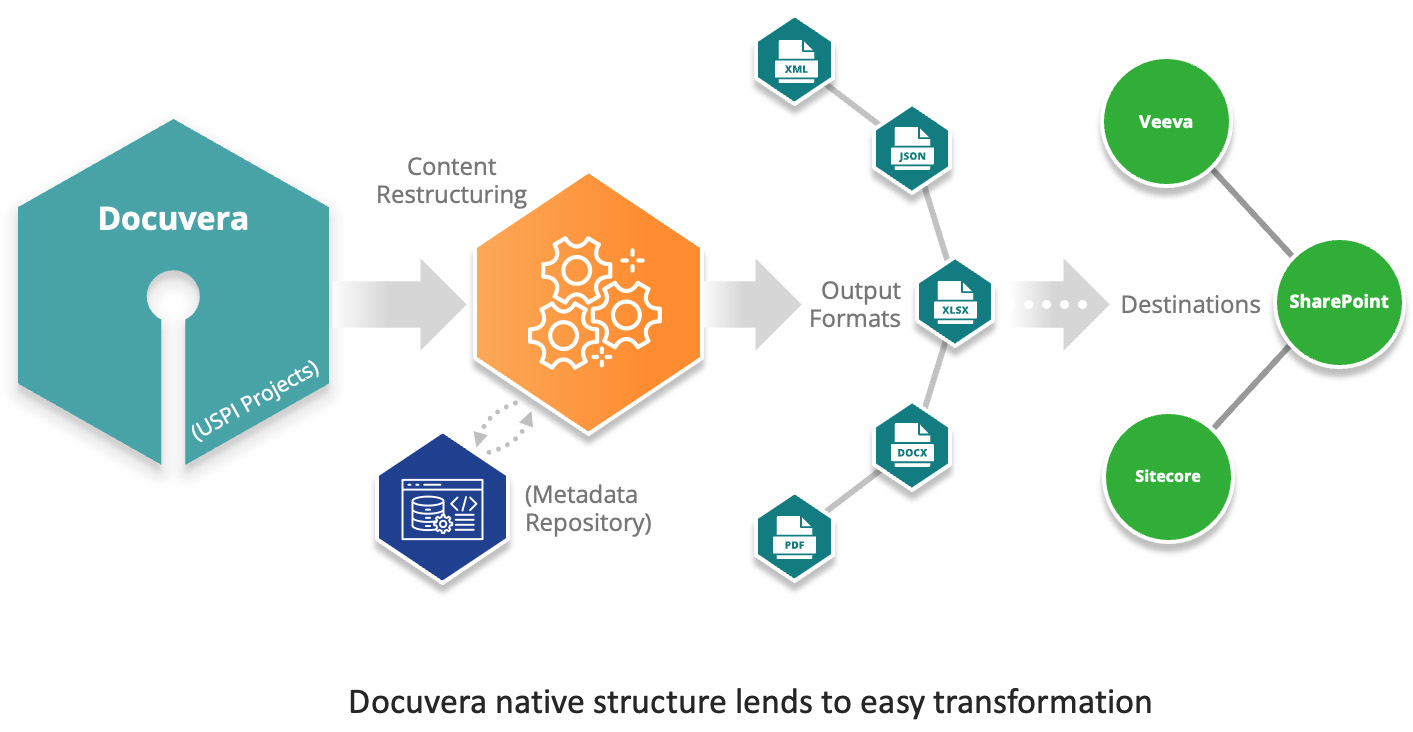
How Docuvera Supports Structured ePI Content
Docuvera is designed to store content in JSON format, separating it from styling elements. This structured approach ensures adaptability across different schemas, enabling users to meet evolving regulatory standards like FHIR and ePI Types I, II, and III, as well as newly emerging frameworks such as those adopted by the Jordan Food and Drug Administration (JFDA) and Health Canada’s electronic labeling initiatives.
Key Features of Docuvera for ePI Compliance:
Schema Flexibility
- Content is stored in a structured, adaptable format that evolves with industry regulations.
- Ensures seamless transitions to updated frameworks
Regulatory-Ready Exports
- Docuvera projects can be exported to match specific regulatory requirements while maintaining content integrity.
- Mapping and relationship management preserve document cohesion and traceability as it moves through compliance workflows.
Advanced Metadata & Tagging
- Docuvera is compatible with most Regulatory Information Management Systems (RIMs) to enable advanced and curated metadata lists to be accessible as content is being created and modified.
- Docuvera handles Metadata and tagging in a structured manner and can facilitate advanced mapping logic to meet ePI needs with ease.
- Ensures that regulatory submissions flow smoothly and return with structured feedback.
Secure & Compliant Transfers
- Docuvera enables system-to-system communication for safe, structured data exchanges between pharmaceutical companies and regulatory bodies.
- Supports interoperability with global healthcare information systems.
Why Structured Content Authoring plays a critical role in ePI compliance
Structured content is more than an efficiency tool—it is a compliance necessity. Utilizing Docuvera, organizations can:
- Ensure regulatory compliance with evolving ePI standards
- Improve content accuracy, traceability, and reusability
- Enhance automation for faster, more efficient regulatory submissions
- Future-proof content for new digital health innovations
As the industry shifts toward structured, machine-readable content, the cost of maintain manual processes for document transformation (either internally or through a third party) will continue to rise. Docuvera provides the technology needed to stay ahead of compliance requirements while future proofing digital healthcare content management.
Contact us today to learn more about how Docuvera accelerates ePI compliance.



