Development Safety Update Report (DSUR)
What is DSUR?
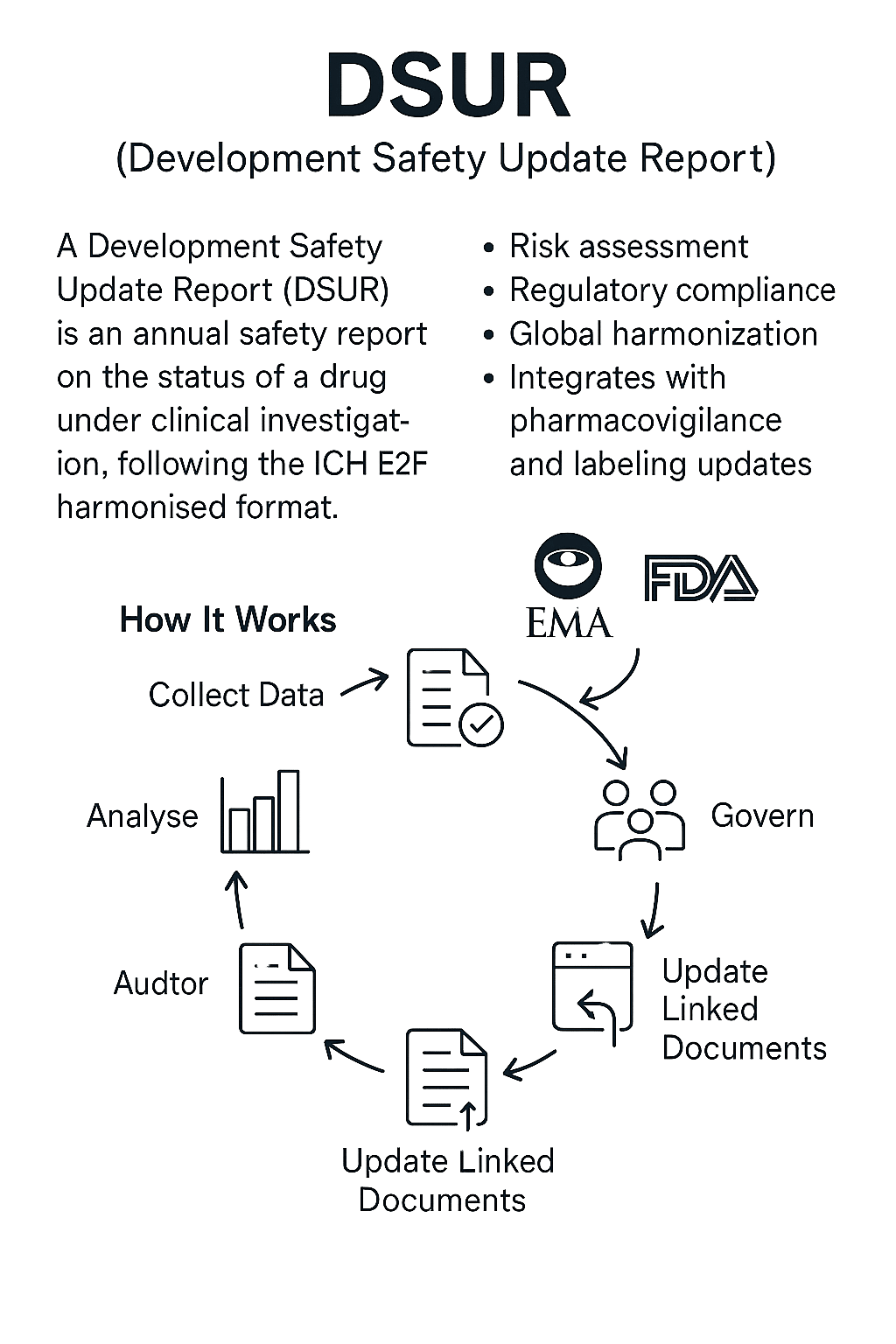
The Development Safety Update Report (DSUR) is an annual safety report required for clinical trial sponsors to provide regulators with a comprehensive assessment of the safety profile of an investigational drug.
It is a globally harmonised format established under the ICH E2F guideline, combining the various regional annual safety reporting requirements into a single, structured report.
The DSUR ensures that regulators receive consistent, complete safety information on investigational products, helping to safeguard trial participants and inform regulatory decision-making.
Why it Matters in Pharma Today
- Global harmonisation — Replaces disparate regional reporting formats with a single ICH-aligned standard.
- Regulatory compliance — Required by multiple regulatory agencies (FDA, EMA, PMDA, Health Canada, etc.) for ongoing trials.
- Continuous safety monitoring — Provides an annual, cumulative review of all available safety data.
- Integration with pharmacovigilance — DSUR data feeds into labeling governance (CCDS, SmPC, USPI, ePI) and informs post-marketing risk management.
- Structured content readiness — Component-based authoring enables reuse of DSUR safety narratives and data tables in other regulatory documents.
Global Landscape
| Region/Body | Governing Entity | Adoption Status / Timeline |
| Global | ICH E2F | Maintains DSUR standard. |
| US | FDA | DSUR accepted as alternative to IND Annual Report. |
| EU | EMA | DSUR required for ongoing clinical trials under Clinical Trials Regulation. |
| Japan | PMDA | DSUR format accepted; submission timelines aligned with ICH guidance. |
| Canada | Health Canada | Accepts DSUR format in place of separate annual safety reports. |
How the DSUR Works in a Component-Based Authoring Model
Collect data — Aggregate all clinical trial safety data from the reporting period, including serious adverse events (SAEs), adverse drug reactions (ADRs), and emerging safety signals.
Analyse — Perform cumulative analyses of safety trends, exposure data, and benefit–risk balance.
Author — Draft DSUR sections according to ICH E2F format, using structured safety tables and reusable narrative components.
Govern — Internal review by clinical, safety, and regulatory teams to ensure accuracy and compliance.
Submit — Provide the DSUR to each relevant regulatory authority within the required timelines.
Update linked content — Use DSUR findings to update CCDS, trial protocols, investigator brochures (IBs), and other safety communications.
DSUR Core Sections (per ICH E2F)
- Introduction
- Worldwide Marketing Approval Status
- Actions Taken for Safety Reasons
- Changes to Reference Safety Information (RSI)
- Estimated Exposure and Demographic Summary
- Presentation of Safety Data (SAEs, ADRs, etc.)
- Significant Safety Findings from Clinical Trials
- Safety Findings from Other Studies
- Safety Findings from Non-Interventional Studies
- Literature Reports
- Lack of Efficacy in Controlled Trials
- Late-Breaking Information
- Overall Safety Assessment
- Summary of Important Risks
- Conclusions
DSUR in Practice
- Emerging liver toxicity signal detected during phase 3 trial → included in DSUR → prompts RSI update in IB and CCDS → cascades to all applicable labels and informed consent forms.
- Unexpected SAE pattern in elderly patients → DSUR assessment leads to protocol amendment to adjust inclusion criteria.