Electronic Product Information (ePI)
What is ePI?
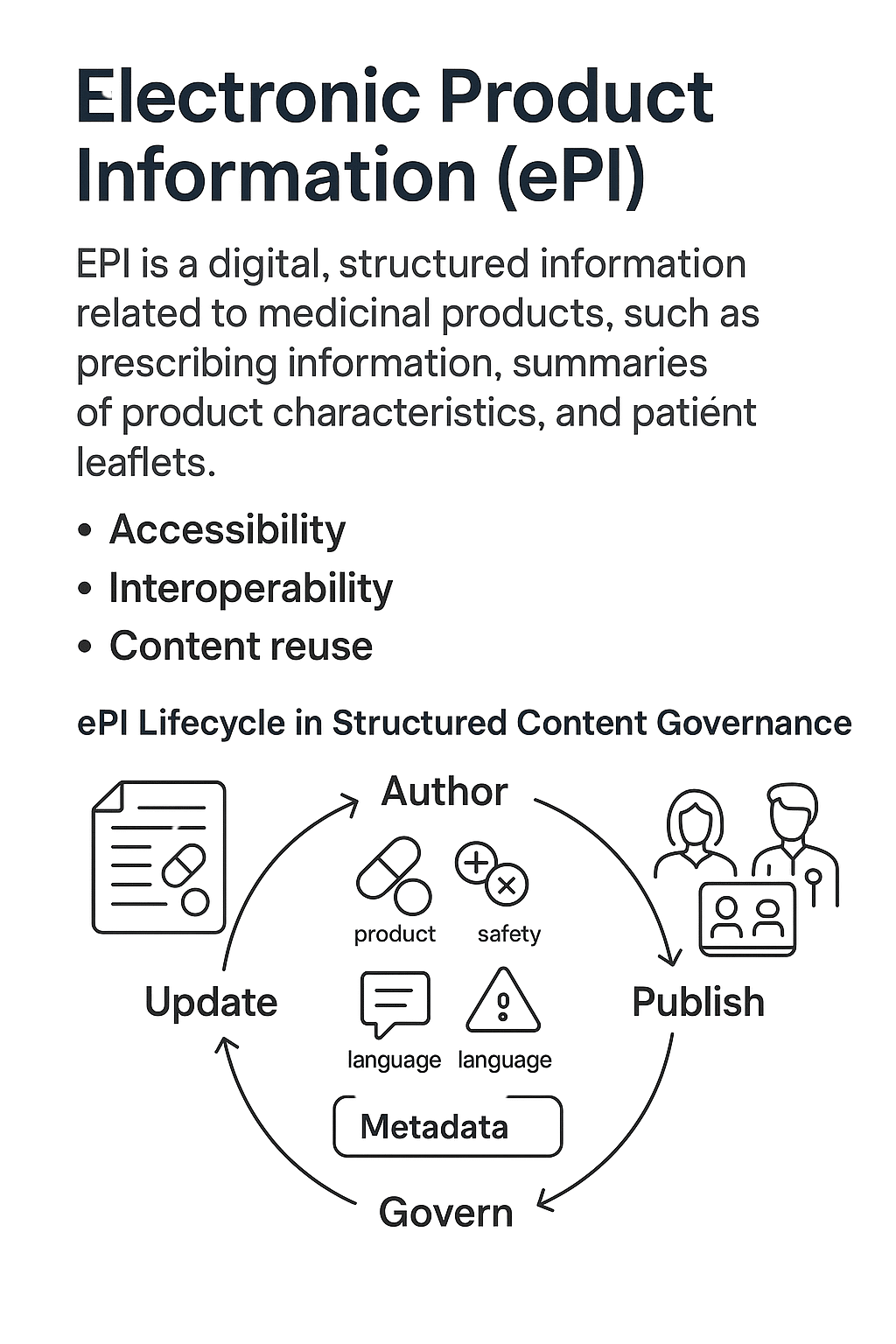
Electronic Product Information (ePI) refers to the digital, structured format of medicinal product information — such as prescribing information, summaries of product characteristics, and patient leaflets — that can be accessed electronically by regulators, healthcare providers, and patients.
Unlike static PDFs, ePI uses structured content and metadata so that information can be searched, filtered, and reused across channels and geographies.
Why it Matters in Pharma Today
- Regulatory push: Agencies like the European Medicines Agency (EMA) and U.S. Food and Drug Administration (FDA) are advancing ePI to improve accessibility, transparency, and update speed.
- Interoperability: ePI formats align with international standards (e.g., HL7 FHIR) for seamless exchange between regulatory, clinical, and commercial systems.
- Patient empowerment: Enables up-to-date, device-friendly content for patients and healthcare professionals in multiple languages.
- Content reuse: Structured ePI content can be propagated into regional formats (e.g., CCDS, SmPC, USPI) without reauthoring.
Global Landscape
| Region/Body | Governing Entity | Adoption Status / Timeline |
| EU | EMA | EU ePI Common Standard |
| US | FDA | Structured Product Labeling (SPL) |
| Japan | PMDA | e-labeling pilot programs aligned with international standards |
How ePI Works in a Component-Based Authoring Model
Source Data — Core product information authored in structured XML/JSON using metadata tags.
Governance Layer — Rules ensure compliance with region-specific formats and update triggers.
Publishing Layer — Content automatically rendered to regulator portals, patient apps, HCP systems.
Feedback & Change Management — Regulatory updates trigger targeted changes across impacted components.