Investigator’s Brochure (IB)
What is the
Investigator’s Brochure?
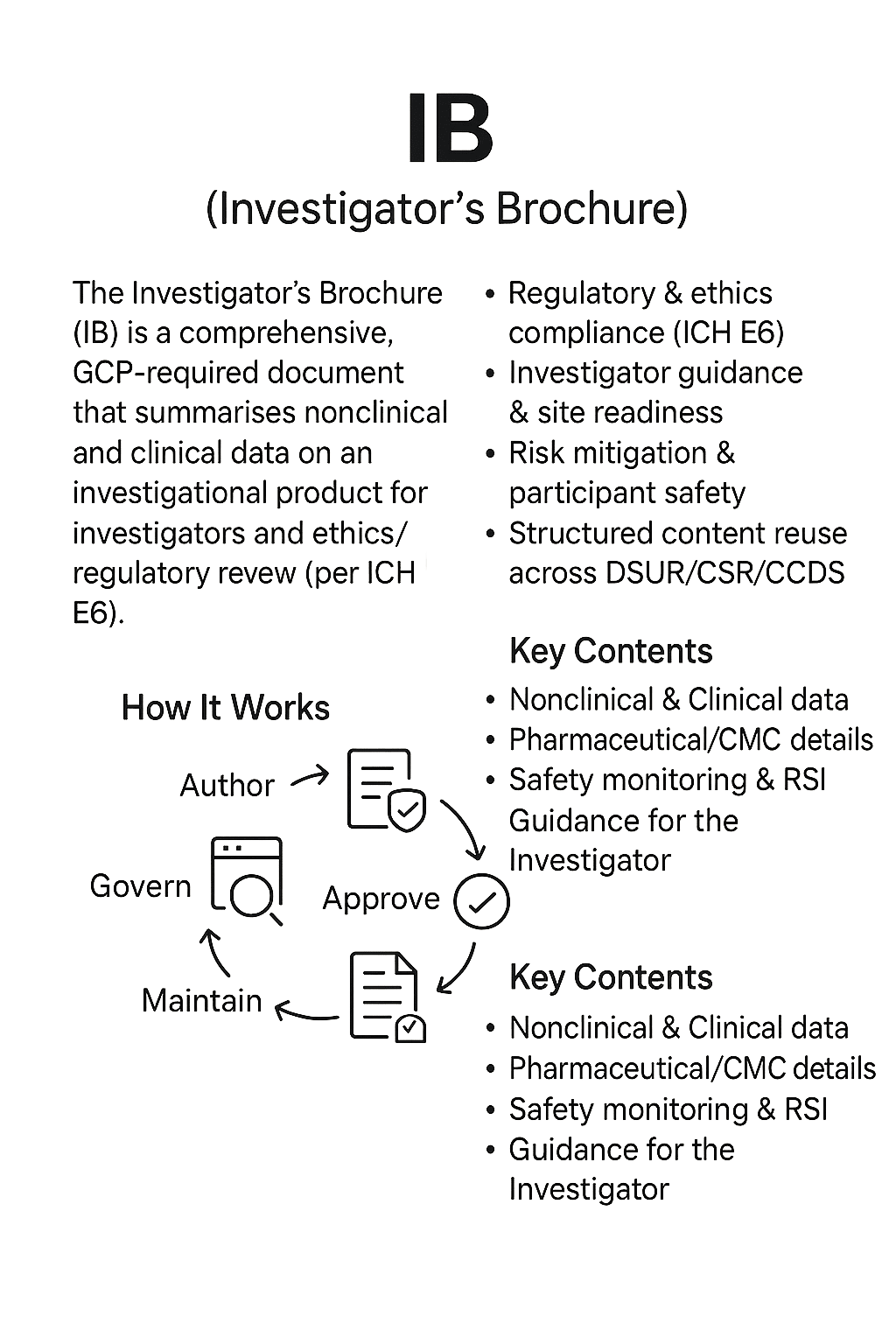
The Investigator’s Brochure (IB) is a comprehensive regulatory document that summarises the clinical and non-clinical data about an investigational product to provide investigators and trial staff with the information they need to conduct clinical studies safely and effectively.
It is a core requirement under ICH E6 Good Clinical Practice (GCP) guidelines and is submitted to regulatory authorities and ethics committees before a clinical trial begins, as well as updated during the trial when significant new information becomes available.
The IB serves as the primary reference for investigational product characteristics, safety profile, and study rationale.
Why it Matters in Pharma Today
- Regulatory compliance — Required by regulators and ethics committees worldwide before and during clinical trials.
- Investigator guidance — Ensures that all investigators have the same comprehensive understanding of the investigational product’s profile.
- Risk mitigation — Facilitates informed decision-making by investigators to protect participant safety.
- Lifecycle linkages — Information from the IB feeds directly into Development Safety Update Reports (DSURs), Clinical Study Reports (CSRs), and ultimately the Core Company Data Sheet (CCDS).
- Structured content opportunity — In a component-based authoring environment, IB sections can be maintained as reusable content, ensuring alignment across all trial documentation.
Global Landscape
| Region/Body | Governing Entity | Adoption Status / Timeline |
| Global | ICH E6 (GCP) | Defines IB content and update requirements. |
| US | FDA | IB reviewed as part of IND submissions. |
| EU | EMA & National Competent Authorities | IB required under Clinical Trials Regulation for authorisation. |
| Japan | PMDA | IB required as part of clinical trial notification process. |
How the IB Works in a Component-Based Authoring Model
Author — Assemble data from preclinical and clinical studies, manufacturing details, and safety monitoring systems into a structured template.
Govern — Review content with cross-functional teams (clinical, regulatory, safety, nonclinical).
Approve — Finalise IB for inclusion in regulatory submissions and ethics committee packages.
Publish — Distribute controlled copies to all investigators and trial sites; maintain in a secure, version-controlled environment.
Maintain — Update IB promptly when significant new efficacy or safety information emerges, ensuring all sites are informed.
IB Sections (per ICH E6)
- Title Page & Confidentiality Statement
- Table of Contents
- Summary
- Introduction
- Physical, Chemical, and Pharmaceutical Properties and Formulation
- Nonclinical Studies
- Effects in Humans (clinical pharmacology, efficacy, safety)
- Summary of Data and Guidance for the Investigator
- References
Example: IB in Practice
- New preclinical toxicity finding → IB updated → notification sent to all investigators → protocol amendments considered if necessary.
- New clinical efficacy data → IB updated to reflect expanded understanding of benefit–risk profile.