United States Prescribing Information (USPI)
What is the USPI?
The United States Prescribing Information (USPI) — also referred to as the “Full Prescribing Information” — is the FDA-approved, legally binding document that provides healthcare professionals in the United States with the essential information needed for the safe and effective use of a prescription drug.
It is part of the product’s New Drug Application (NDA) or Biologics License Application (BLA) approval and must conform to the FDA’s Physician Labeling Rule (PLR) format.
The USPI serves as the official source of truth for product characteristics, dosing, administration, contraindications, warnings, and other regulatory-approved information.

Why it Matters in Pharma Today
- Regulatory compliance — A USPI must be reviewed and approved by the FDA before a product can be marketed in the US, and any subsequent changes require FDA approval via supplements.
- Clinical decision-making — It provides healthcare providers with authoritative guidance to inform prescribing, administration, and patient monitoring.
- Legal and liability protection — Serves as the legally recognised statement of a product’s approved uses and risks; deviations can carry significant legal consequences.
- Integration with digital health systems — Forms the basis of structured product labeling (SPL) XML files used in electronic health records and pharmacy systems.
- Structured content alignment — In a component-based authoring environment, USPI sections can be linked directly to CCDS and SmPC content for global consistency.
Global Landscape
| Region/Body | Governing Entity | Adoption Status / Timeline |
| United States | Food and Drug Administration (FDA) | Mandatory for all prescription drugs and biologics; must meet PLR requirements. |
| Global Context | WHO, ICH | USPI content structure aligns in part with international labeling harmonisation efforts but remains US-specific. |
| Comparative Labeling | EMA / EU | USPI is comparable to the EU SmPC but with distinct format, section order, and content rules. |
How the USPI Works in a Component-Based Authoring Model
Author — Draft USPI content based on the CCDS and US-specific regulatory guidance.
Govern — Internal medical, legal, and regulatory review to ensure compliance with PLR formatting and FDA guidance documents.
Approve — Submit to FDA for review as part of an NDA/BLA or as a supplemental application for updates.
Publish — Make available in print and electronically via DailyMed and other FDA-approved channels; generate SPL XML for structured data use.
Maintain — Update promptly for new safety information, indications, dosing changes, or manufacturing updates, ensuring alignment with CCDS and other global labels.
Structure of the USPI (per PLR format)
- Highlights of Prescribing Information (summary table)
- Full Prescribing Information (FPI)
- 1. Indications and Usage
- 2. Dosage and Administration
- 3. Dosage Forms and Strengths
- 4. Contraindications
- 5. Warnings and Precautions
- 6. Adverse Reactions
- 7. Drug Interactions
- 8. Use in Specific Populations
- 9. Drug Abuse and Dependence (if applicable)
- 10. Overdosage
- 11. Description
- 12. Clinical Pharmacology
- 13. Nonclinical Toxicology
- 14. Clinical Studies
- 15. References
- 16. How Supplied / Storage and Handling
- 17. Patient Counseling Information
Example: USPI in Practice
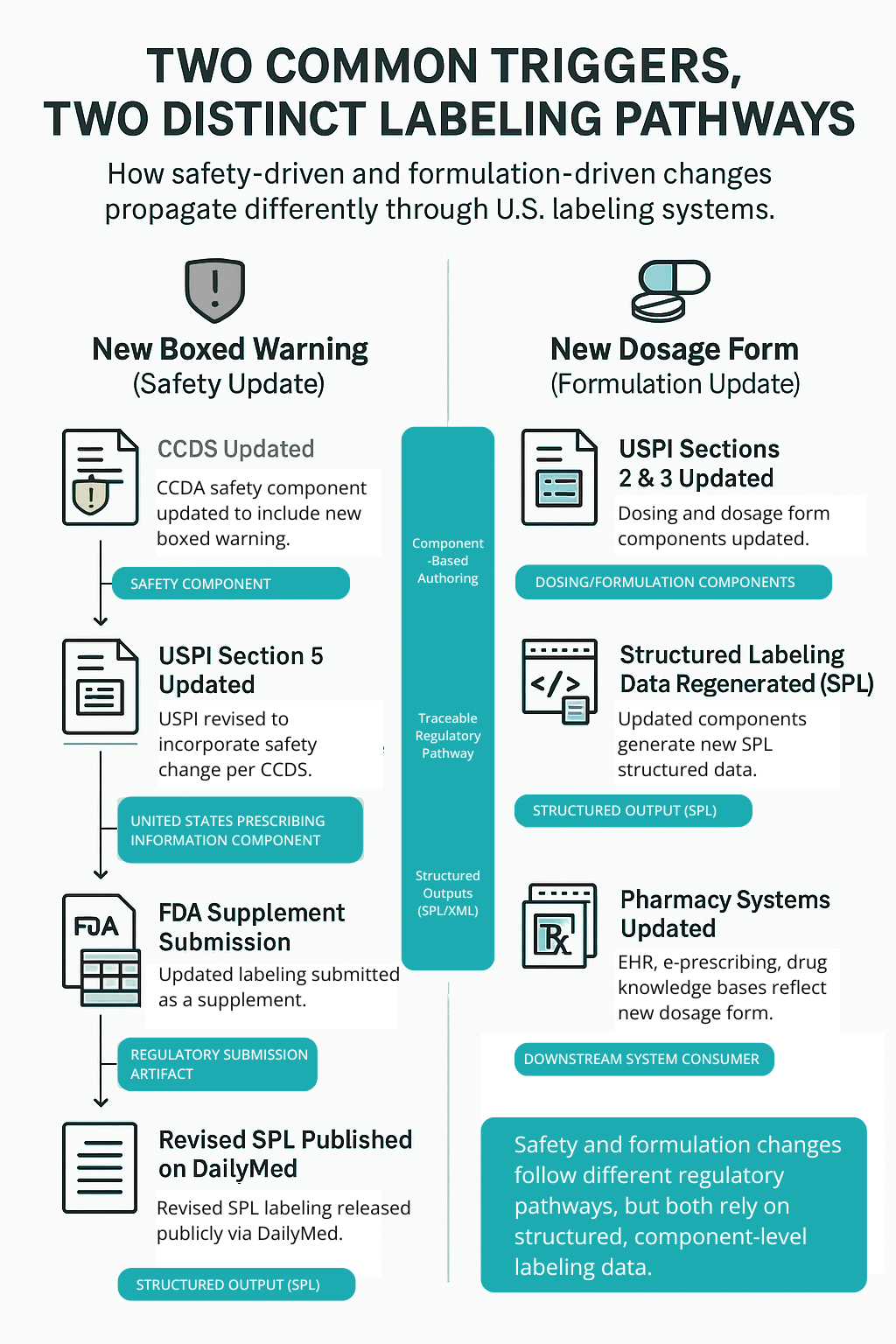
- New boxed warning — CCDS updated → USPI Section 5 updated → FDA supplement submission → revised SPL XML published on DailyMed.
- New dosage form — USPI Sections 2 and 3 updated → pharmacy systems receive updated structured labeling data.