There is a specific kind of deadline that does not announce itself with urgency. It arrives quietly on a roadmap, gets noted in a Management Board summary, and then one day it is not optional anymore. EMA’s rollout of electronic product information, which includes the EMA ePI requirements, is that kind of deadline.
At its March 2026 Management Board meeting, EMA confirmed the phased go-live for ePI across centrally authorised products. Vaccines and oncology products come first, with phased CAP go-live beginning from Q4 2026. EMA also states that, once the new EU pharmaceutical legislation enters into application, ePI will be mandatory for all newly authorised medicines, highlighting the EMA ePI requirements. The voluntary period is closing. What replaces it is a structural requirement with recurring operational consequences.
The question for regulatory affairs and labeling teams now is not whether ePI is coming. It is whether their content infrastructure is positioned to absorb the EMA ePI requirements.
Understanding the EMA ePI Requirements for Compliance
Electronic product information is not a reformatted PDF. EMA defines ePI as the authorised statutory product information for medicines — including the summary of product characteristics, package leaflet, and labelling — adapted for handling in electronic format and dissemination via the web, e-platforms, and print. The EU ePI Common Standard and its FHIR implementation approach specify how that content must be structured, coded, and expressed at the data level rather than the document level. Sections, subsections, coded terminology, and multilingual content are defined as structured content elements, not as static page layout.
EMA’s ePI pilot, which ran from July 2023 to August 2024 across Denmark, the Netherlands, Spain, and Sweden, resulted in the publication of 23 ePIs from real regulatory procedures and showed that the EU system can move toward phased implementation, while still requiring additional functionality, IT integration, and business-process development. Multilingual content management remains one operational challenge. Cross-market consistency remains another. Both persist without structural solutions at the authoring level.
The implication is practical. EMA’s roadmap states that once a product’s PI is available in electronic format, it will remain electronic in all subsequent variations. For organizations that author in Word or static document formats, that means a conversion step at every one of those events unless structured output is generated from the source. That cost is not absorbed once at implementation. It recurs across the entire submission lifecycle.

Why the CMC Convergence Matters Here
ePI does not arrive as an isolated requirement. In parallel, PQ/CMC initiative is developing structured CMC submissions using HL7 FHIR, while ICH M4Q(R2) is part of a broader push toward more efficient lifecycle management, digitalisation, and knowledge management in quality submissions. Labeling and manufacturing documentation are not the same workstream, but they are moving in the same structured-data direction at the same time.
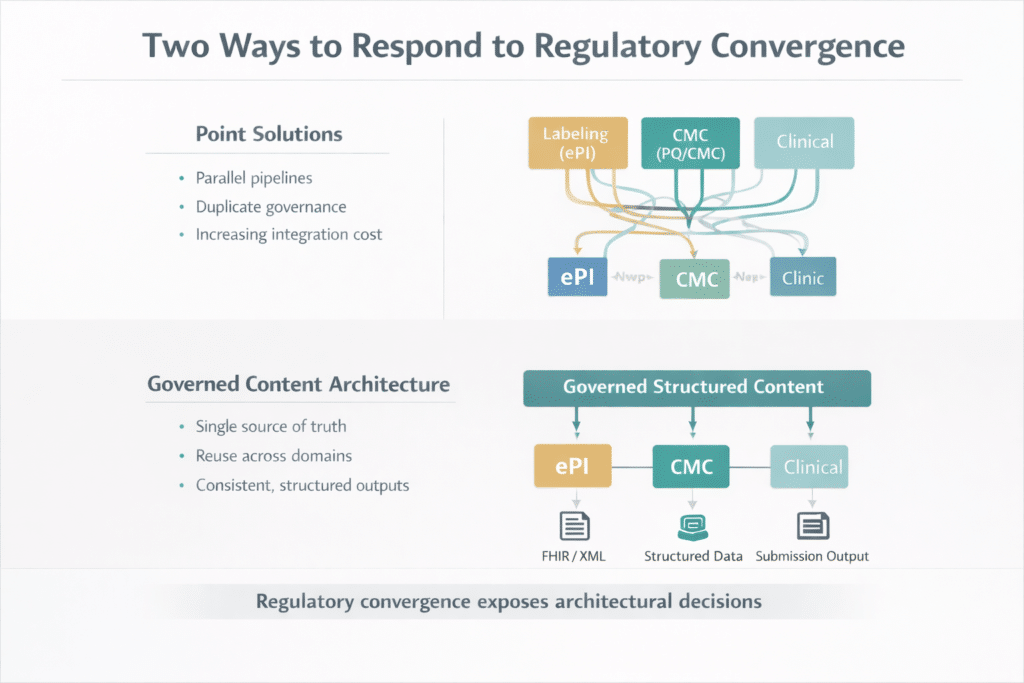
Organizations that treat ePI as a standalone labeling project will solve one problem while leaving the conditions that create it intact. A conversion pipeline for product information, built alongside unchanged CMC workflows, produces two content architectures to maintain, two sets of transformation rules to validate, and two separate governance models to audit. The operational question is not whether to address these mandates. It is whether to address each one with a point of solution, or to build a governed architecture that accommodates all of them.
The regulatory direction is consistent. EMA ePI is explicitly built around the EU ePI Common Standard and FHIR-based implementation. FDA’s PQ/CMC work is explicitly being developed as an HL7 FHIR implementation guide for structured Module 3 data exchange. At the same time, quality submissions are moving toward more granular, digital, lifecycle-oriented organization. Each point solution adds integration cost. Each requirement addressed within a unified structured content architecture reduces it.
The Authoring Decision at the Center of This
EMA’s standard specifies structure at the data level. Authoring in that format from the source removes the conversion step entirely. A global labeling manager whose team works from structured, governed content components does not treat ePI as a remediation project. They configure a rendering format.
That distinction matters operationally because EMA’s draft roadmap says applicants will be requested to author and upload ePI at the PLM portal as an additional process step alongside the current Word/PDF submission during initial implementation.
The alternative — extracting, converting, validating, and reconciling content from static documents at each submission cycle — compounds across portfolios. A single centrally authorized product with annual variations and periodic renewals generates multiple conversion events per year. Across a portfolio of 20 or 30 products, the aggregate cost is not marginal.
This is also where the persona of the Global Labeling Manager intersects directly with the decision. Persona 3 in our ICP framework — the Functional User and Domain Steward — is the person who carries this operational burden daily. They manage CCDS, SmPC, and USPI content across dozens of markets, where a single safety update can cascade into 40 or more country-specific revisions. The efficiency gain from authoring structured content once, at the source, is not abstract for this person. It is measurable in review cycles avoided, reconciliation hours recovered, and variation submissions completed without remediation overhead.
For teams in this position, Docuvera’s structured content authoring platform is built specifically for this class of problem. Content is authored once in governed, structured components. Metadata is controlled at the element level. FHIR-based ePI output is generated from a single authoritative source. When a new output format is required, it is a rendering configuration, not a content rebuilding exercise. The governance layer ensures traceability, version control, and audit readiness across every lifecycle event.
Docuvera provides structured content authoring software for regulated content teams. This article is intended as operational analysis of current regulatory direction, not legal advice.
That operating model also fits the phased implementation logic in EMA’s roadmap: initial CAP implementation is English first, with all languages optional at first and fuller multilingual rollout later.
For a deeper examination of the ePI compliance architecture and what it requires from labeling operations across labeling and CMC workflows, see The New Risk Surface in the Pharma Content Lifecycle.
The Timeline Is Set
Vaccines and oncology products are first in EMA’s phased CAP rollout from Q4 2026. Other therapeutic areas follow in subsequent phases. EMA states that, once the new pharmaceutical legislation enters into application, ePI will be mandatory for all newly authorised medicines.
The organizations that begin content architecture investment now will reach that deadline with systems already generating compliant output. The organizations that wait will arrive with a remediation project and a deadline. The difference shows up in every submission cycle that follows.
Frequently Asked Questions:
Sources
- EMA Management Board: Highlights of March 2026 Meeting — ema.europa.eu
- Electronic Product Information, EMA — ema.europa.eu/epi
- EU ePI Common Standard — build.fhir.org
- ICH Quality Guidelines (M4Q) — ich.org [5] FDA PQ/CMC Initiative — fda.gov
- ePI FHIR Implementation Guide — epi.ema.europa.eu/fhirig/