Regulatory data silos are a strategic problem hiding in plain sight in pharma
In pharmaceutical organizations, regulatory data silos in pharma are rarely identified as a top strategic risk. It does not usually appear on transformation roadmaps or executive dashboards. Yet it quietly undermines two outcomes every pharma leader cares about: speed and trust. Fragmented data does not cause dramatic failures overnight. Instead, it introduces small inefficiencies and inconsistencies that accumulate until they surface as delayed launches, regulatory friction, or loss of confidence in execution.
The scale and complexity of pharmaceutical companies make data management a persistent challenge. Fragmented regulated data across regulatory, labeling, safety, and adjacent clinical functions creates friction that slows coordination, complicates reuse, and makes it harder to maintain a consistent view of approved information across markets. In practice, that friction shows up as more reconciliation, more clarification, and less confidence in what should be authoritative data. See the EMA Substance and Product Data Management Services overview.
Most organizations experience fragmentation because of how work is structured. Regulatory, labeling, clinical, safety, and affiliate teams all interact with the same underlying information, but they do so through different systems, processes, and timelines. Over time, data becomes duplicated across documents, repositories, and regional variants. Each copy feels necessary in the moment. Collectively, they create systemic risk, especially when organizations still rely on disconnected document workflows.
Why regulatory data silos become the default operating model in pharma
Fragmentation emerges naturally in complex, global organizations. Teams optimize locally to meet deadlines and regulatory requirements. Affiliates adapt global information to local contexts. External partners maintain their own working versions. These behaviors are rational responses to pressure, but they result in decentralized ownership of what should be shared data.
Once multiple copies exist, alignment depends on human diligence. Updates must be communicated, interpreted, and applied consistently across every instance. As portfolios grow and timelines compress, this manual coordination becomes increasingly fragile. Manual processes are required to keep fragmented data aligned, but these workflows are labor-intensive and error-prone. Fragmentation is no longer an exception; it becomes the operating norm.
This is why many organizations experience a growing gap between how data should behave and how it actually behaves in practice. That gap becomes more visible as regulatory operations scale across markets and data has to move through increasingly digital submission and review processes. See FDA Electronic Regulatory Submission and Review.
Organizations struggle to retrieve and validate siloed data efficiently, making it harder to bring the right approved information to the right decision-maker at the right time.
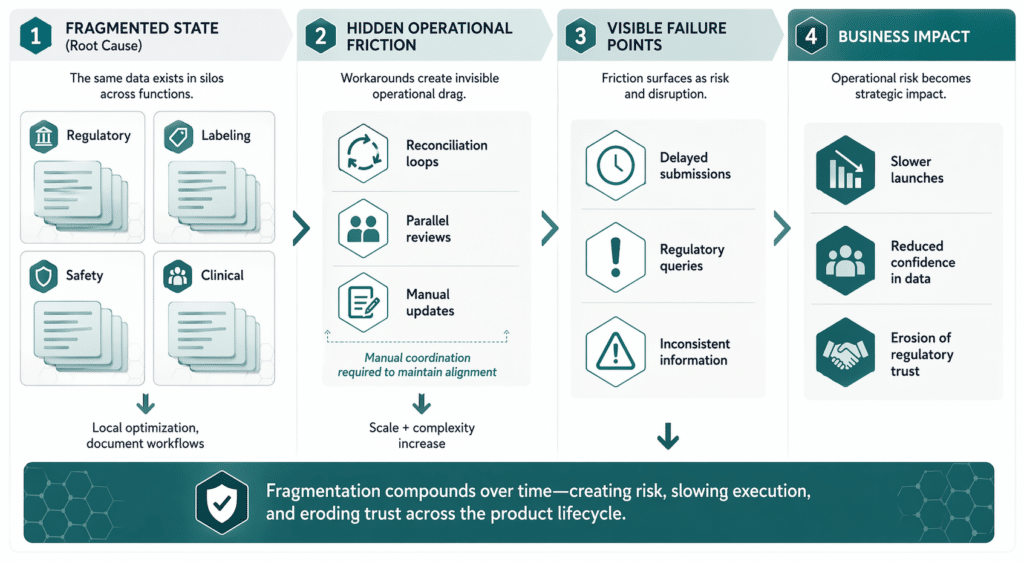
Inconsistency is structural, not accidental
When data is fragmented, inconsistency is inevitable. The same fact may be phrased slightly differently in different contexts. Timing differences emerge as updates are applied unevenly. Local adaptations persist longer than intended. None of this reflects carelessness. It reflects systems that allow divergence to occur silently.
Regulators are acutely sensitive to these inconsistencies. A safety statement that appears differently across submissions or markets raises questions, even if the underlying science is unchanged. Queries are often framed as requests for clarification, but they signal uncertainty about which version of the data is authoritative. Accuracy becomes paramount in these scenarios, because consistency, reliability, and traceability across regulatory uses are central to compliant execution. See EMA Substance and Product Data Management Services.
This uncertainty has consequences. Reviews slow. Confidence erodes. Teams are forced into reactive explanation mode instead of proactive execution. Without integration, teams risk missing critical signals in an increasingly noisy environment. FDA’s Electronic Regulatory Submission and Review framework and eCTD standard reinforce how formalized and format-dependent regulated submission environments already are.
The operational cost of reconciliation and rework
Inside the organization, fragmented data creates a heavy operational burden. Experts spend time comparing documents, tracing changes, and explaining differences. Parallel review cycles multiply effort. Escalations become common when alignment cannot be established quickly.
These activities do not advance submissions or improve quality. They exist solely to compensate for misaligned data foundations. The cost is paid repeatedly, across every submission, variation, and update. These reconciliation and rework activities consume valuable resources and reduce overall efficiency, diverting attention from innovation and strategic initiatives.
Over time, this hidden work absorbs capacity that could otherwise be focused on higher-value activities.
Fragmentation is expensive long before it becomes visible. Investing in the right capabilities can reduce operational burden and improve the speed and quality of regulated work.
Why fragmented regulated data erodes regulatory trust
Trust with regulators is built on consistency and traceability. Regulators expect the same information to remain consistent wherever it appears in the submission and review process. When data fragments across documents, systems, or markets, that expectation is weakened—not always through error, but often through ambiguity. See FDA Electronic Regulatory Submission and Review.
Each inconsistency, however minor, introduces doubt. Each clarification request consumes time and attention. Over time, interactions become less predictable. Organizations may find themselves subject to increased scrutiny, not because of scientific concerns, but because data behavior appears uncontrolled.
Trust is not lost in a single moment. It erodes incrementally as fragmentation accumulates. With a solid data foundation, companies can support more consistent execution and more reliable reuse of approved information at scale. See EMA SPOR master data and EMA Substance and Product Data Management Services.
How centralized regulatory data management improves speed, consistency, and trust
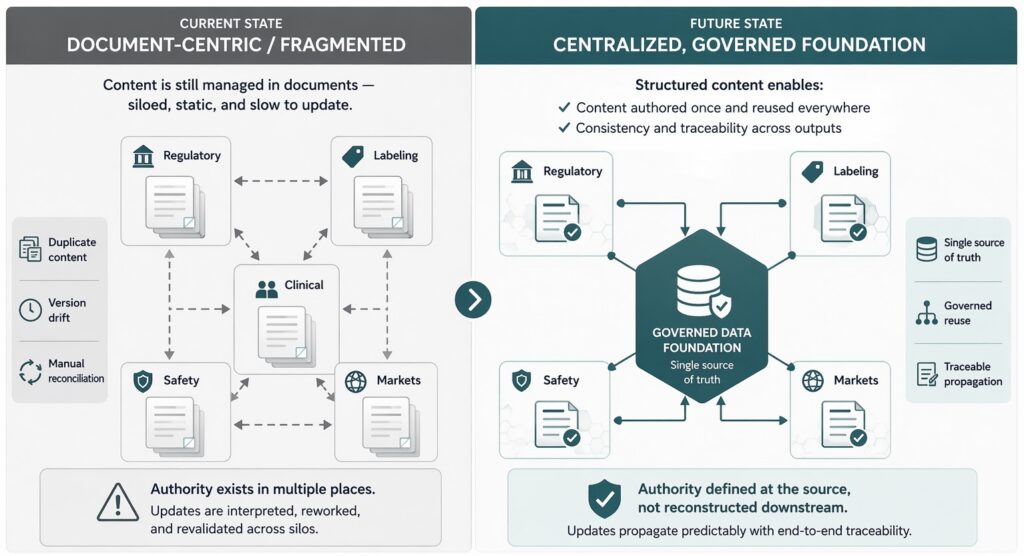
Centralized data management addresses fragmentation at its source. By establishing a single authoritative foundation for regulated data, organizations eliminate the conditions that allow divergence to persist. All downstream use draws from the same approved information. Differences become intentional and governed, rather than accidental and hidden. Enabling better collaboration and integrating data across functions helps teams work from a shared, governed source, improving consistency and reducing downstream reconciliation. See EMA SPOR master data.
Centralization does not mean eliminating local flexibility. It means anchoring flexibility to a shared truth. Local adaptations are managed as variations within structured product information requirements, not independent copies. Alignment becomes a property of the system, not a heroic effort by individuals. Integrating data across functions improves visibility and makes decision-making, reuse, and risk identification more reliable across the product lifecycle.
This shift fundamentally changes how organizations operate. Reuse becomes safe. Updates propagate predictably. Confidence replaces caution, and platforms like Docuvera can accelerate this process.
How Docuvera eliminates fragmentation at the source
Docuvera is designed to champion centralized data management in environments where fragmentation is common and costly. Instead of allowing regulated information to proliferate across documents and systems, Docuvera maintains a single, governed data foundation that supports enterprise-wide reuse.
By anchoring data centrally, Docuvera ensures that regulatory, labeling, and operational teams are always working from the same authoritative source. Divergence becomes visible immediately, not discovered downstream. Docuvera gives regulatory, labeling, and operational teams a governed foundation for using the same approved information consistently across functions and markets. Governance and traceability are embedded, reducing the need for manual reconciliation and revalidation.
Importantly, Docuvera does not require organizations to abandon existing systems or workflows. It complements them by ensuring that the underlying data remains aligned as it moves across functions and markets. This makes harmonized regulated data easier to reuse, review, and maintain across evolving submission and lifecycle workflows
The compounding benefit of eliminating fragmentation
When fragmentation is addressed, benefits accumulate quickly. Submission timelines can shorten because less effort is spent reconciling versions and explaining differences. Regulatory interactions improve because information aligns more consistently. Post-approval updates also become easier to manage because data does not need to be rediscovered or reassembled each time.
Equally important, organizational confidence increases. Teams trust the data they are using. Decision-making accelerates. Escalations decline. The organization moves from reactive correction to proactive control.
These gains are not one-time improvements. They compound as the data foundation strengthens. Harmonizing clinical data improves data quality, consistency, and analytical capabilities.
Fragmentation is optional — but only with the right foundation
Fragmented data is not an inevitable consequence of scale or regulation. It is the result of architectures that allow duplication and divergence to persist. Organizations that invest in centralized, governed data management remove a major source of hidden friction from their operations.
Docuvera provides the foundation required to make this shift sustainable. By eliminating fragmentation at the source, it enables organizations to move faster, build trust, and operate with greater confidence across the entire product lifecycle. As regulatory operations become more digital and data-dependent across the US and EU, integrated, high-quality data becomes more important to speed, consistency, and control. A strong data foundation supports more reliable execution across the product lifecycle.
The hidden cost of fragmented data is paid every day in lost time, strained relationships, and missed opportunity. Organizations that recognize and address it gain an advantage that extends far beyond individual submissions.
Learn more about how Docuvera champions data centralization by exploring our governance layer or global labeling solutions, or by reaching out to our team.
Learn more about how Docuvera champions data centralization by reaching out to our team.