Digital transformation is not a system problem — it is a data flow problem
Digital transformation initiatives often center on which platforms to deploy or retire. In practice, transformation succeeds or fails based on how data flows across the ecosystem those platforms create. This data flow is fundamental to achieving successful outcomes.
In pharma, regulatory data integration pharma only creates value when it supports authoritative, governed data flow across that hybrid environment. Without reliable, governed data movement, new technology simply digitizes existing inefficiencies.
Speed does not emerge from modernization alone; it emerges when data can move predictably, authoritatively, and interoperably across organizational boundaries. That requires a durable source of truth for regulated information, not just more connected applications.
That is why regulatory data integration pharma — not legacy system replacement — is the real unlock for digital transformation. Additionally, embracing legacy system integration in pharma is essential for optimizing workflows and enhancing data accuracy.
Legacy system integration in pharma is critical to ensuring that data can be accessed and utilized effectively across various platforms. Emphasizing legacy system integration in pharma allows organizations to maximize the value of their existing infrastructure while meeting regulatory demands.
Moreover, it is pivotal in aligning disparate data sources, enhancing accuracy, and streamlining workflows across teams.
This is why platforms like Docuvera are increasingly evaluated not on feature parity with legacy systems, but on whether they can serve as a stable data foundation that allows structured, regulated data to move across the enterprise without being redefined at every handoff. This direction aligns with current regulatory modernization efforts, including the FDA’s Pharmaceutical Quality/Chemistry, Manufacturing and Controls (PQ/CMC) structured data program and the European Medicines Agency’s Substance, Product, Organisation and Referential (SPOR) master data services.
Why pharma cannot — and should not — rip and replace legacy systems instead of integrating them
Through integrating legacy systems pharma organizations can foster better collaboration and communication between departments, ensuring that critical information flows seamlessly.
Pharma operates under constraints that make wholesale system replacement both risky and unrealistic. Regulatory information management (RIM) systems, EDMS platforms, safety databases, and quality systems are deeply embedded in validated GxP processes. They represent years of configuration, inspection history, and institutional knowledge.
Replacing them outright introduces operational and regulatory risk that few organizations can justify.
As a result, most pharmaceutical environments will remain hybrid for the foreseeable future. New platforms will coexist with legacy systems, each serving a specific purpose.
This hybrid reality is not inherently problematic. The challenge arises when data becomes trapped within or fragmented between systems. Teams are often forced to compensate manually to maintain alignment, which can lead to inefficiencies.
That is why regulatory content interoperability and governed data flow across the RIM, EDMS, safety, and quality layer have become more practical objectives than wholesale replacement.
Organizations that succeed in this environment do not attempt to eliminate legacy systems. Instead, they introduce a centralized regulatory data foundation, often through platforms such as Docuvera’s Governance Layer, that allows legacy and modern systems to coexist without fragmenting the underlying information.
The importance cannot be overstated, as it directly impacts the efficiency and effectiveness of regulatory submissions and compliance.
The hidden cost of broken data flow between systems
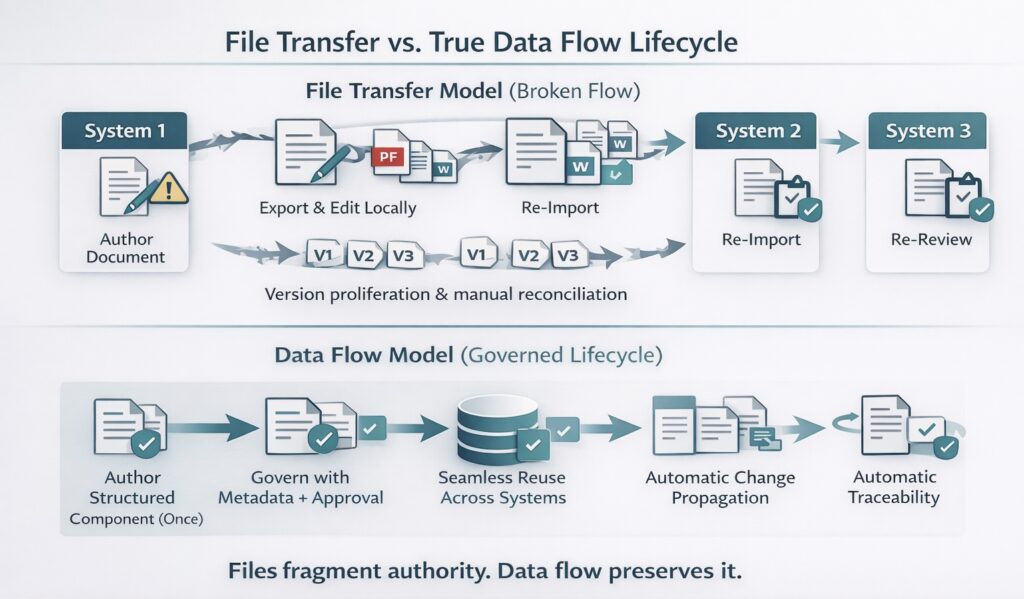
When data does not flow cleanly, organizations rely on workarounds. Documents are exported from one system, edited elsewhere, and re-imported into another.
Teams perform manual comparisons to ensure alignment. Previously approved information is re-reviewed simply because its provenance is unclear. These activities introduce delay, consume expert capacity, and increase the risk of inconsistency.
Ultimately, embracing legacy system integration will lead to a more agile and responsive organization in the face of evolving regulatory requirements.
Crucially, these delays are often misattributed. Timelines stretch not because regulatory requirements are unusually complex, but because data must be rediscovered, reconciled, or revalidated each time it crosses a system boundary.
Regulatory queries increase when information diverges across platforms, even if the underlying content is correct. Regulators increasingly expect data integrity, traceability, and controlled lifecycle management to be maintained continuously, not reconstructed after the fact. The cost of broken data flow is paid repeatedly, across every submission and update.
A shift in pharma towards legacy system integration also opens the door for innovations that leverage data more effectively, driving better outcomes.
What seamless data flow actually means in regulated environments
Seamless data flow does not imply uniformity of systems. Different platforms exist for good reasons: submissions, document control, safety, and quality all have distinct requirements. True regulatory data integration means these systems share a common, authoritative data foundation rather than competing sources of truth. In practice, that foundation behaves more like governed master data than a series of synchronized document copies. That distinction is increasingly relevant in Europe, where EMA’s SPOR services are designed to facilitate the reliable exchange of medicinal product information in a robust and consistent manner through centralized master data management.
In a governed data-flow model, such as Docuvera’s, information is created once and reused many times. Changes propagate predictably. Governance, traceability, and context travel with the data as it moves. Systems consume data according to their role without redefining or duplicating it, which improves submission readiness and reduces reconciliation during dossier preparation and post-approval change activity. This is also the operating logic behind FDA’s PQ/CMC program, which is built on structured data standards aligned with ICH eCTD v4.0 and mapped to HL7 FHIR resources — the same logic applied to how companies should manage their own regulatory data internally. This distinction matters because moving files is not the same as moving data. Files fragment authority; pharmaceutical data flow preserves it.
Why point-to-point integration strategies fail at scale in pharma
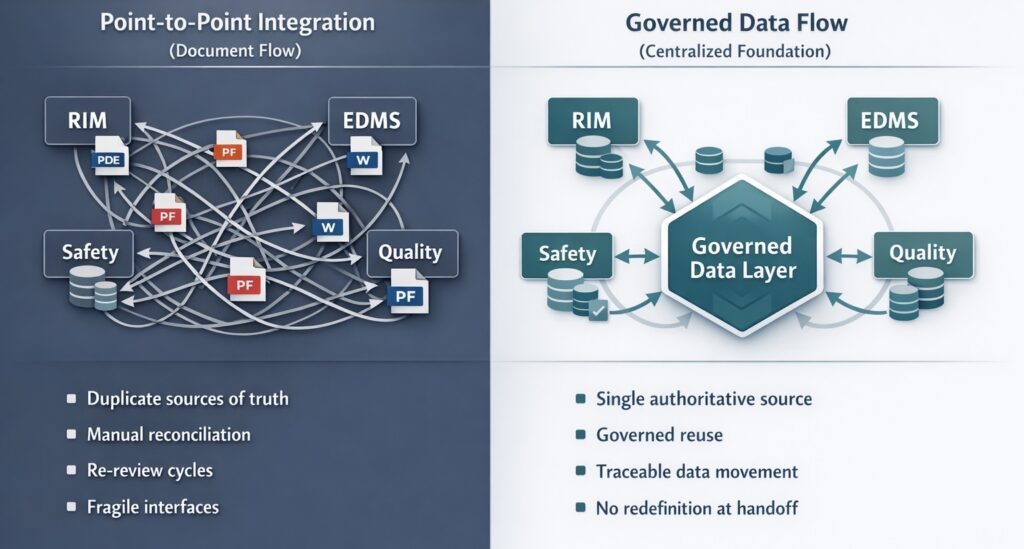
Many integration efforts rely on custom, point-to-point connections between systems. While these may solve narrow problems, they introduce fragility as complexity grows.
Each new interface adds maintenance overhead. Each regulatory change requires rework. Over time, organizations become locked into brittle architectures that are expensive to sustain and difficult to adapt.
Point-to-point integrations treat data as an attribute of systems rather than as an enterprise asset. In these approaches, governance weakens at each boundary. Data becomes tightly coupled to specific workflows instead of being managed independently from a single source of truth. The organization gains connectivity but not coherence.
How Docuvera establishes a centralized data foundation across ecosystems
Docuvera is designed to address this structural issue by acting as a centralized regulatory data foundation for regulated information. Rather than replacing existing RIM, EDMS, safety, or quality systems, Docuvera complements them by managing regulated data independently of where it is ultimately consumed.
In this model, data is authored, governed, and maintained once within Docuvera. Downstream systems draw from that authoritative foundation rather than redefining or duplicating information. This allows existing systems of record to remain in place while reducing the number of competing sources of truth. Governance rules, lineage, and context remain intact as data moves across the ecosystem. Legacy platforms continue to function as systems of record for their intended purpose, but without fragmenting the data landscape. This is the same operating logic that underpins Docuvera’s approach to regulatory compliance and risk mitigation — treating regulatory data as a governed asset rather than a series of disconnected document outputs.
Why centralized regulatory data integration changes operational behavior
The shift from system-centric to data-centric integration alters how teams work. When data is authoritative and shared, re-review becomes the exception rather than the rule. Parallel execution becomes viable because alignment is assumed rather than constantly re-established. This shift leads to a more effective and efficient workflow.
Speed improves not because teams work faster, but because uncertainty is removed. Submission timelines shorten as reconciliation disappears. Regulatory interactions become more predictable as information aligns across contexts. Post-approval updates accelerate because data does not need to be reconstructed for each system or market. This same dynamic underpins Docuvera’s positioning as the cornerstone of digital transformation in regulated environments.
Legacy system integration in pharma is essential for ensuring that data can be accessed and utilized effectively across various platforms, allowing organizations to maximize the value of their existing infrastructure while meeting regulatory demands.
These gains are durable. They scale with portfolio growth because they are grounded in data architecture, not individual effort.
Data flow as the prerequisite for future readiness
The importance of governed data flow extends beyond current operations. Regulatory expectations are steadily moving toward structured, interoperable, and machine-readable information. FDA’s PQ/CMC program is intended to increase review efficiency through structured, standardized submission data, with FHIR profiles aligned to ICH eCTD v4.0, while EMA’s SPOR services support centralized master data and more consistent exchange of medicinal product information in line with ISO IDMP standards. At the same time, organizations are exploring automation and AI to manage complexity. None of these initiatives succeed without clean, authoritative data that can move safely between systems. EMA’s Data Quality Framework reinforces the same requirement from a regulatory decision-making perspective, defining quality criteria across extensiveness, coherence, reliability, timeliness, and relevance for data used in regulatory decisions.
A centralized data foundation—such as the one Docuvera provides—allows organizations to evolve without destabilizing validated environments. New capabilities can be layered on incrementally. Change becomes manageable rather than disruptive, consistent with the lifecycle-oriented quality system expectations described in ICH Q10. This is also why Docuvera emphasizes support for structured labeling and product information standards including FDA SPL, ePI, EMA DADI, and HL7 FHIR and use cases such as global labeling.
Speed follows when data is allowed to move
Digital transformation in pharma is not about minimizing the number of systems in the landscape. It is about removing the invisible barriers that prevent data from moving between them. Organizations that design for seamless, governed regulatory data integration achieve speed, trust, and scalability without increasing risk.
Those that continue to treat integration as file transfer will remain constrained, regardless of how modern their platforms appear. When data flows, speed follows — not as an aspiration, but as an operational outcome of sound data architecture.
In conclusion, effective legacy system integration in pharma is essential for maximizing the value of existing infrastructures and ensuring compliance with regulatory standards.

