Pharma timelines don’t slip because teams are slow, they slip because data is immobile. The regulated information driving global submissions, global labeling, and compliance is still trapped in static documents that cannot flow or adapt at the speed modern science demands. Structured content and data management (SCDM) systems provide a centralized platform for storing, organizing, and analyzing these complex datasets. Pharmaceutical companies generate vast amounts of data throughout the drug development process, resulting in massive datasets that require advanced tools for integration, analysis, and decision-making. For regulatory affairs, CMC writers, labeling managers, and pharmacovigilance leads, the practical consequence is the same: every submission cycle absorbs time that should be spent on science, not reconciliation.
These systems are essential for ensuring data security and compliance with regulatory requirements. Centralized, secure storage involves using secure repositories with strong encryption and access controls to protect sensitive data, such as research findings, patient information, and clinical trial results, from breaches and to ensure regulatory compliance.
Why “Speed” Initiatives Rarely Deliver Lasting Gains
Most life sciences organizations have already optimized their processes. They’ve invested in RIM upgrades, hired submission leads, standardized templates, and digitized approval workflows. But despite this, McKinsey research shows that less than one-third of organizations say their transformations have achieved sustained performance improvement — a pattern that holds in life sciences. Data silos are one of the most significant challenges faced by pharma R&D, leading to inefficiencies and delays.
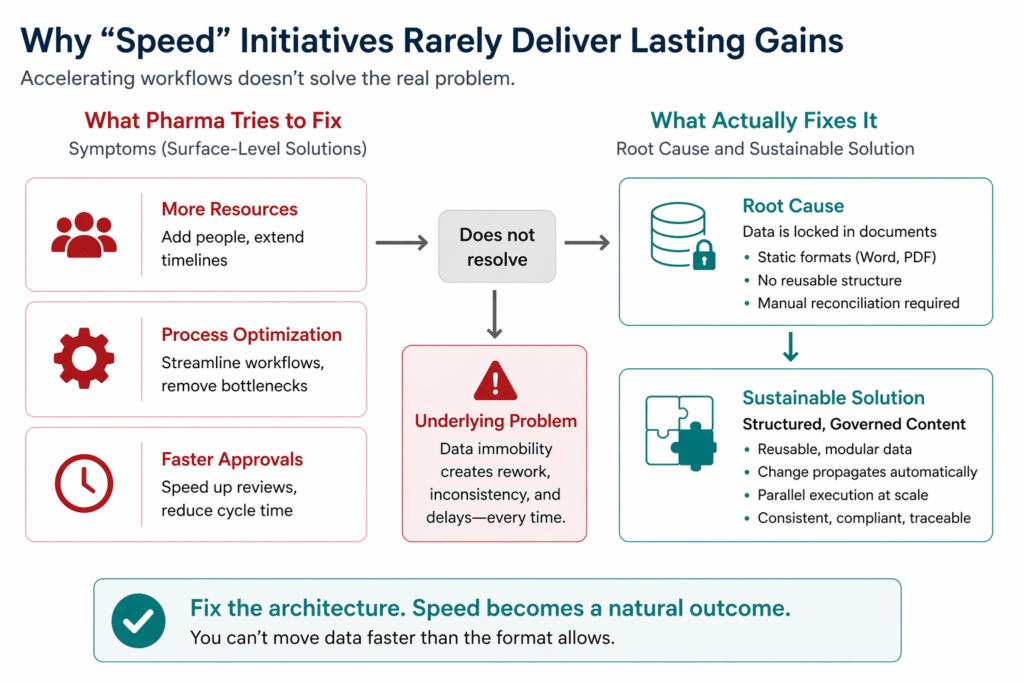
The issue lies in how the industry defines “speed.” Moving faster within a document-based environment is a short-term victory. Every submission still requires human reconciliation of data embedded in Word files, PDFs, or regional versions. Manual tasks required to reconcile data from multiple sources contribute to delays and inefficiencies in regulatory data operations. Documents, by their nature, are static containers. Data cannot move faster than its format allows.
Speed initiatives treat the symptoms: resourcing, bottlenecks, handoffs. But without addressing the immobility of the underlying data, every improvement remains temporary. Maintaining data quality and integrity is crucial for reliable analysis and decision-making in pharma R&D. Best practices in pharmaceutical R&D data management emphasize robust governance, security, standardization, data integration, and adherence to data quality standards.
What Changes When Regulated Content Is Treated as Structured Data
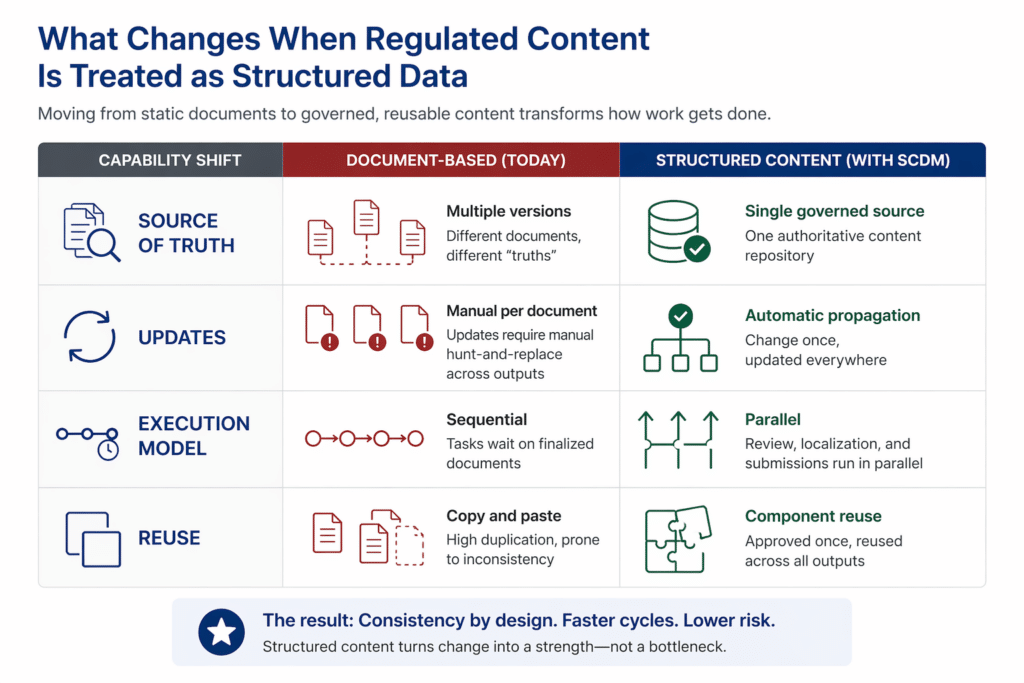
It’s essential to treat regulated information not as document text, but as structured, reusable data. This is the foundation of structured content and data management. Industry researchers from Amgen and Janssen, writing in AAPS Open, define SCDM as a component-based approach that allows information to be modularized and reused across a centrally managed content repository — directly addressing many of the inefficiency challenges in regulatory submissions. In this model, each statement, specification, or claim exists as a governed component—tagged with metadata, approved once, and reused everywhere. Master data management (MDM) is a key approach in this context, centralizing and unifying critical data assets to provide a single source of truth for the pharmaceutical industry.
Docuvera’s research and customer outcomes show that when data is created and managed this way, organizations can achieve up to 80% content reuse across submissions and a 20% acceleration in overall cycle times.
This shift transforms the entire operational model:
- Single authoritative data sources: Instead of multiple versions of “truth,” all content—clinical, CMC, labeling—draws from a unified, validated repository, with data governance ensuring consistent classification, documentation, and control of data assets. This process incorporates quality assurance and ensures data accuracy, both of which are critical for maintaining data integrity and regulatory compliance.
- Change propagation instead of document reconciliation: When a component is updated, that change cascades automatically to every dependent output. For labeling teams managing CCDS, SmPC, USPI, and ePI variants, this eliminates the manual hunt-and-replace work that consumes update cycles.
- Parallel execution instead of sequential dependencies: Review, localization, and submission preparation can proceed simultaneously, rather than waiting for finalized documents. Regulatory affairs, CMC, and pharmacovigilance functions can work against the same validated component library without blocking one another.
By leveraging MDM, organizations enable data-driven decisions, reduce inefficiencies, and accelerate bringing therapies to market.
The implications extend far beyond compliance. This data-centric foundation enables automation, interoperability, and readiness for the AI-driven future of content assembly and validation.
Time-to-Market Is a Data Flow Problem Across the Drug Lifecycle
When viewed through this lens, time-to-market becomes a data flow problem, not a workflow problem. Every phase of the product lifecycle depends on information movement:
- Initial Submissions: Structured content enables faster dossier assembly by drawing directly from approved data blocks rather than rewriting narratives, streamlining regulatory submissions including CMC content for Module 3 and ensuring accurate trial data from clinical trials is included for compliance and transparency.
- Variations and Updates: Instead of reauthoring multiple versions, teams update a single governed component, ensuring consistency across markets and products. This is where regulatory content reuse delivers compounding returns over a product’s lifecycle.
- Global Rollouts: With interoperable, machine-readable content (FHIR, IDMP, SPL), regional affiliates can localize and publish updates in days, not months, supporting the drug development process and meeting evolving regulatory requirements. Docuvera’s global labeling platform demonstrates this in practice across CCDS, SmPC, USPI, and ePI workflows.
- Drug Development Lifecycle: Pharma data management spans the entire drug development lifecycle, from initial research and clinical trials to manufacturing, supply chain, and post market surveillance. Effective management of patient data at each stage is critical for regulatory compliance and, most importantly, for patient safety.
Data management systems in pharma support collaboration and communication among stakeholders, such as researchers, clinicians, regulators, and patients. These systems help companies comply with regulations by implementing robust security measures, access controls, and audit trails. They enable researchers and clinicians to analyze data more efficiently, identify patterns and trends, and make evidence-based decisions. Effective pharmaceutical data management ensures information is accurate, accessible, and compliant with regulations like FDA Good Clinical Practice guidelines, GDPR, and HIPAA. A unified platform for data management can accelerate innovation in drug development by improving decision-making and data accessibility.
This lifecycle-wide alignment is what transforms “speed” from a one-time win into a repeatable capability. It’s not about doing the same things faster—it’s about doing different things altogether.
Regulatory authorities are already accelerating this shift. The European Medicines Agency’s EU ePI Common Standard, based on HL7 FHIR, establishes structured, machine-readable submissions as the foundation for harmonized electronic product information across the EU, and the FDA’s Structured Product Labeling (SPL) standard is expanding to new submission types across CDER, CBER, CDRH, CVM, and OTC products. These mandates confirm that structured, data-driven authoring is no longer optional—it’s foundational to future compliance.
How Docuvera Operationalizes Structured Content and Data Management
Docuvera was purpose-built for this new paradigm. Unlike retrofitted document management systems, Docuvera enforces structure, governance, and interoperability at the point of creation. Each content component—whether a dosing instruction or manufacturing statement—carries its metadata, approval status, and lineage.
This approach ensures that:
- Regulated data is centralized into a governed, authoritative foundation that connects regulatory, clinical, and labeling domains. Implementing a centralized data system allows for a significant reduction in clinical trial costs and manufacturing downtime.
- Updates made once are reflected everywhere that data appears, ensuring traceability and eliminating redundant rework. This is the operational backbone behind ePI readiness across regional regulatory variants.
- Teams work in parallel, drawing from the same validated source rather than duplicating effort across regions or functions. Regulatory writers, CMC authors, labeling managers, and pharmacovigilance leads operate against one governed component library rather than competing document silos.
- AI operates safely, within a governed framework that preserves auditability and compliance, supported by robust security measures, access controls, and audit trails to protect sensitive information and ensure regulatory adherence. Automated data observability tools enable real-time monitoring and remediation of data anomalies before audits.
Master data management (MDM) helps pharmaceutical companies comply with regulatory requirements by implementing robust security measures and audit trails. Complementary technologies such as blockchain are also being explored to create tamper-proof audit trails for clinical trial data and product serialization.
The results are measurable. Docuvera customers report that the platform has reduced re-review cycles, accelerated labeling updates, and saved millions annually through avoided rework and earlier product availability.
The Strategic Takeaway: Sustainable Speed and Data Quality Come from Structure
Pharma doesn’t have a productivity problem—it has a data architecture problem. As long as critical information remains authored in static, document-based formats, pharma time to market will remain constrained by manual reconciliation and version control.
Sustainable acceleration comes only from structure. By treating content as governed data—modular, traceable, and machine-readable—organizations unlock agility across every function. Regulatory, clinical, and manufacturing teams can collaborate on the same information backbone, with confidence that each update is consistent, compliant, and globally aligned.
As Docuvera’s work with leading life sciences organizations has shown, when governance moves upstream and data is treated as a living asset rather than a static deliverable, “speed” stops being a temporary initiative and becomes a permanent capability. In this model, time-to-market improvements are not the product of heroics—they are the outcome of design.
Driving innovation and achieving a competitive advantage in pharma data management depend on operational efficiency and robust data management practices. In this environment, “speed” is not an act of effort but a property of architecture. When data moves freely, submissions accelerate, updates synchronize automatically, and compliance becomes proactive rather than reactive. The industry’s next leap forward won’t come from more people or more process—it will come from data that can finally move. AI integration into pharmaceutical data management is reshaping how companies approach data processing, quality assurance, and decision-making across all drug development phases.