Global scale amplifies labeling and data alignment problems before it amplifies value
Global expansion is one of the most powerful growth levers in the pharmaceutical industry. Each new market extends patient reach, revenue potential, and portfolio value. At the same time, global scale dramatically increases operational complexity. In this environment, organizations often discover that their biggest constraint is not regulatory diversity, but global data misalignment. That challenge becomes especially visible in global labeling harmonization, where teams must keep core product information, local variations, and downstream outputs aligned across markets without slowing execution.
Global data misalignment refers to instances where data across an organization’s departments is fragmented, inconsistent, or tells conflicting stories, leading to slow, unconfident, or incorrect business decisions. When information behaves inconsistently across markets, speed declines and risk increases regardless of how capable local teams may be.
Why global execution exposes labeling and data weaknesses
At a local level, teams can often compensate for imperfect data foundations. Manual coordination, institutional knowledge, and informal communication help bridge gaps. At global scale, these compensations collapse. The volume of interactions overwhelms human coordination.
Core regulatory and product data is reused across dozens of markets, often simultaneously. Safety updates, labeling changes, and lifecycle variations must propagate quickly and consistently. When data is duplicated across documents, systems, and affiliates, alignment becomes fragile. Minor differences escalate into significant delays as teams scramble to reconcile information across regions.
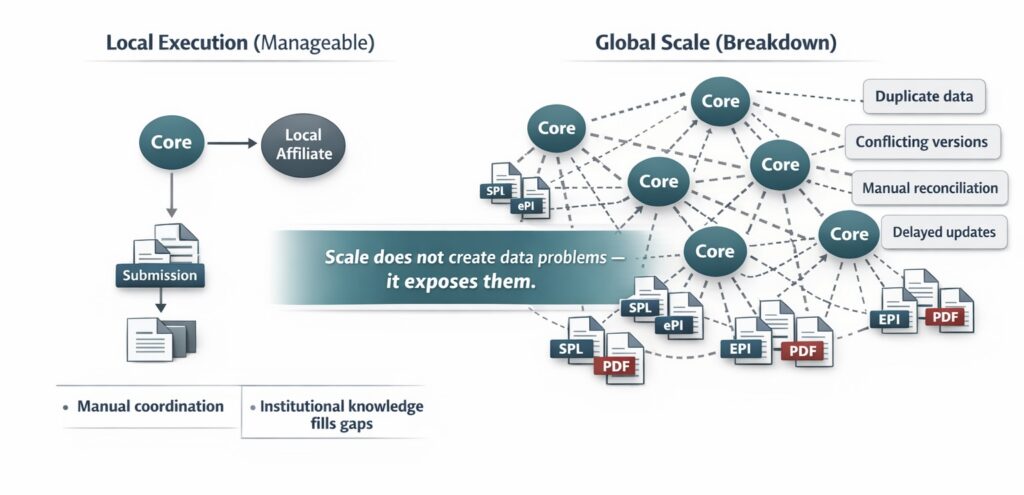
Global scale does not create data problems—it reveals them. Regulators are also moving toward more structured, digital, and reusable product information models. In the EU, EMA and its partners are enabling the use of electronic product information (ePI) for human medicines, while in the US, FDA has adopted Structured Product Labeling (SPL) as a mechanism for exchanging product and facility information.
The local copy trap and its consequences
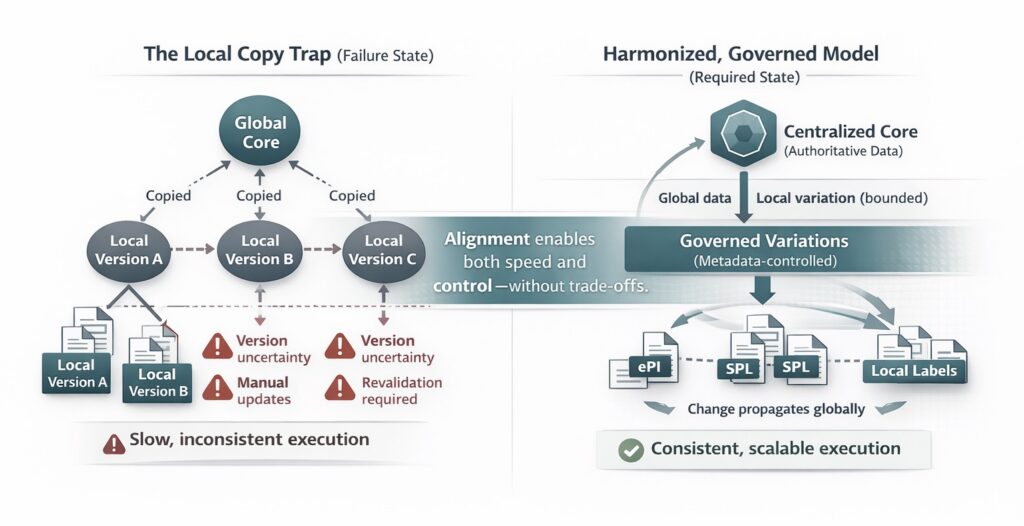
To meet local timelines, affiliates often create their own working copies of global information. This behavior is rational under pressure, but it introduces long-term risk. Local copies drift from the global core. Updates are applied unevenly. Variations persist beyond their intended scope.
The local copy trap is a common and costly pattern in global pharma operations. It transforms scale from an advantage into a drag on execution.
Over time, the organization loses confidence in its own data. Global teams are unsure which version is current. Local teams are unsure when updates apply. Coordination slows as alignment must be re-established manually for each change.
Why harmonization, not standardization, is the real goal
Effective global execution does not require rigid standardization. Markets differ, and local context matters. What global organizations need is harmonization, not uniformity. Alignment ensures that shared facts remain consistent while controlled variation is applied intentionally.
This distinction is critical. When alignment is absent, organizations are forced to choose between speed and control. When alignment is present, they achieve both. Local teams can move quickly because they trust the global core. Global teams can update confidently because they know changes will propagate correctly and predictably.
Alignment must be designed into the data architecture. It cannot be sustained through coordination, governance meetings, or heroics alone.
Centralized data as the backbone of global labeling harmonization
Centralized data management provides the backbone for alignment at scale. By maintaining a single authoritative foundation for regulated data, organizations ensure that all markets draw from the same source of truth. Local adaptations are governed as variations rather than independent artifacts.
Centralization makes dependencies visible. It clarifies which data elements are global, which are local, and how changes affect downstream outputs. Updates can be applied once and reflected consistently across regions, reducing the need for manual synchronization and revalidation. Teams moving toward structured product information are better positioned to make those dependencies explicit across affiliates, submissions, and formats.
Without centralized data alignment, global execution inevitably slows as portfolios and markets expand.
How Docuvera enables global scale through global labeling harmonization
Docuvera is designed to support global pharma organizations by championing centralized, governed data management. Instead of allowing core regulatory data to fragment across affiliates and systems, Docuvera maintains a single authoritative data foundation that supports controlled local use.
Within Docuvera, global core data is defined, governed, and reused across markets. Local variations are managed transparently, ensuring that differences are intentional, traceable, and compliant. When updates occur, their downstream impact across regions is immediately visible, enabling faster and more confident execution.
Docuvera complements existing regional workflows and systems rather than replacing them. Regulatory, labeling, and operational platforms continue to function as designed, but they draw from aligned data. This preserves local agility while eliminating the alignment tax that typically accompanies global scale.
Global alignment as a prerequisite for future growth
Regulatory expectations are evolving toward greater transparency, faster updates, and more digital-first interaction models. That direction is becoming more explicit as regulators and industry groups move toward digital, structured product information models such as ePI in the EU and SPL in the US. At the same time, organizations are investing in automation and AI to manage complexity. These initiatives depend on aligned, trustworthy data foundations.
Global data alignment enables organizations to adapt without destabilizing operations. New requirements can be absorbed without re-architecting workflows. Innovation becomes more scalable when organizations can maintain controlled, traceable operations consistent with ICH Q10 Pharmaceutical Quality System.
Data alignment is not just an operational concern—it is a strategic prerequisite for sustained global growth.
Scale rewards organizations that harmonize labeling and align data first
Global pharma growth rewards organizations that address alignment before complexity overwhelms execution. Those that rely on coordination alone will slow as scale increases. Those that invest in centralized, governed data foundations gain speed, confidence, and resilience.
Docuvera provides the infrastructure required to align data at scale, enabling organizations to grow globally without slowing down. When data is aligned, scale becomes an advantage rather than a liability.