The false tradeoff between compliance and speed
Pharmaceutical organizations have long assumed that compliance and speed exist in tension. The prevailing belief is that stronger controls inevitably slow execution, while faster execution increases risk. This assumption has shaped operating models for decades, reinforcing review-heavy processes, sequential approvals, and layers of quality checks designed to catch issues late in the lifecycle.
As pharma portfolios grow, markets expand, and regulatory expectations increase, this model is breaking down. Reviews multiply, timelines stretch, and teams struggle to keep pace…not because they lack discipline, but because the system is overloaded. The problem is where and how pharma data governance is applied. Regulatory expectations around data integrity and lifecycle control have only reinforced that pressure in regulated environments, as reflected in the FDA’s data integrity guidance and the EMA’s guidance on data integrity across the data lifecycle
The organizations that move fastest today are not those that relax controls. They are those that shift governance upstream, embedding it directly into how data is created, managed, and reused.
Why governance breaks down at scale
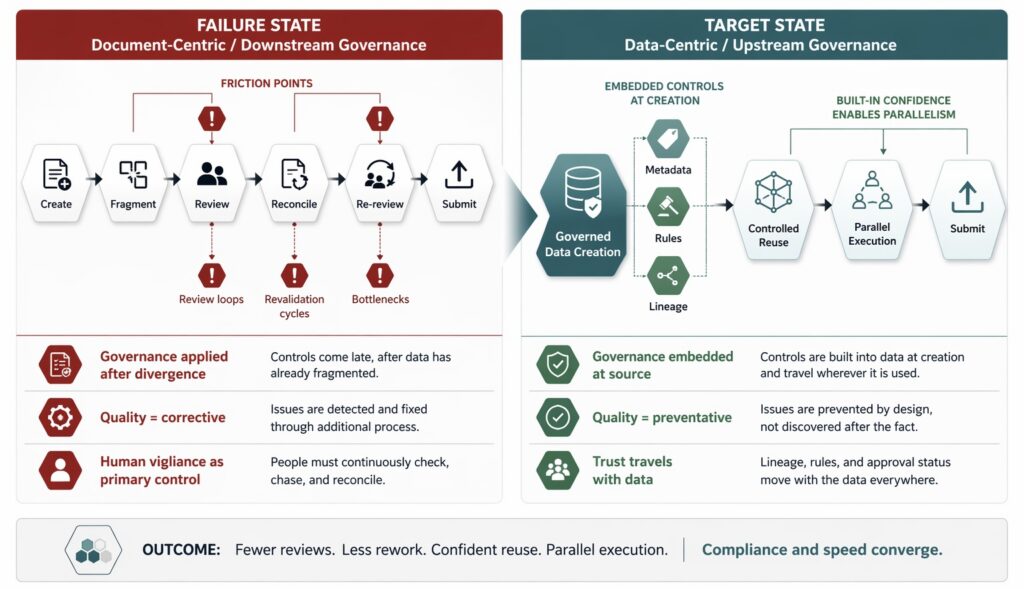
Traditional governance models rely heavily on people and process. Data is created in documents, passed through workflows, and reviewed repeatedly to ensure alignment. Quality control acts as a corrective layer, catching inconsistencies after they appear. Human vigilance becomes the primary control mechanism.
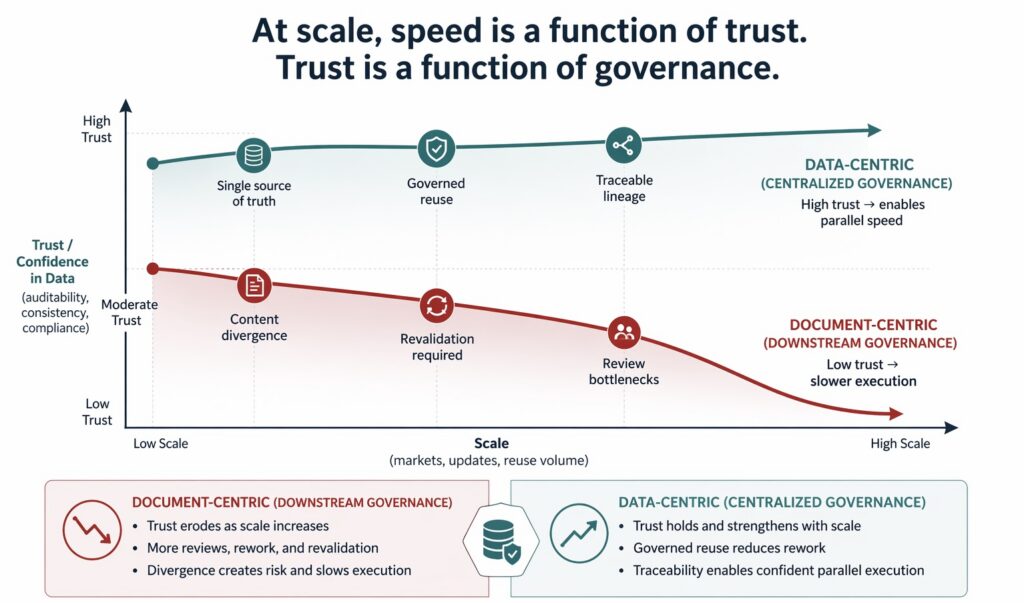
This approach can work at small scale. At enterprise scale, it fails. As the volume of data increases, the number of markets grows, and update cycles accelerate, manual enforcement becomes brittle. Reviews turn into bottlenecks. Revalidation becomes routine. Teams spend more time confirming alignment than advancing work.
This breakdown is not caused by carelessness. It is structural. When governance is applied downstream—after data has already fragmented—speed and compliance both suffer. This misalignment directly conflicts with regulator expectations that data integrity, traceability, and control be maintained throughout the data lifecycle, not reconstructed retrospectively, a principle reinforced in both the FDA’s data integrity Q&A and the European Medicines Agency’s data integrity guidance.
Data-centric governance changes the operating model
Centralized data governance reverses this dynamic. Instead of relying on repeated review to enforce consistency, rules are embedded directly into the data itself. Constraints are applied at the point of creation. Reuse rules define how data can be deployed across contexts. Lineage and ownership are explicit.
This is also why data-centric operating models are increasingly linked to structured content and governed reuse in pharmaceutical regulatory work: when content is modular, centrally managed, and traceable, organizations reduce repetitive verification and improve consistency across submissions and lifecycle updates.
In this model, compliance becomes preventative rather than corrective. Issues are avoided by design, not discovered after the fact. The burden shifts from constant verification to controlled reuse.
This shift has profound implications for speed. When data is governed at its source, teams can reuse it confidently without revalidation. Parallel execution becomes viable because alignment is assumed. The organization moves faster precisely because uncertainty has been removed.
Governance as an enabler of execution, not a brake
When governance is embedded upstream, it no longer acts as a brake on execution. Instead, it enables momentum. Teams spend less time seeking confirmation and more time progressing work. Escalations decline because discrepancies are resolved at the data level, not through interpretation.
This is particularly important in regulated environments where the cost of ambiguity is high. Clear governance reduces the need for conservative, sequential processes designed to compensate for uncertainty. Speed emerges as a byproduct of clarity.
Why centralized data governance matters
Centralization is essential to making data-centric governance work at enterprise scale. Without a single authoritative data foundation, rules cannot be enforced consistently. Data fragments across systems and documents, and governance reverts to manual oversight. Quality-system expectations also reinforce the need for lifecycle-wide consistency and demonstrable control.
Centralized data governance, as an operating model rather than a tooling decision, ensures that:
- there is one source of truth,
- governance rules apply uniformly,
- reuse is safe and intentional,
- changes propagate predictably.
This does not mean eliminating flexibility. It means anchoring flexibility to a shared, governed core.
How Docuvera operationalizes data-centric governance
Docuvera is designed to champion centralized data governance in complex, regulated environments. Rather than treating governance as an external process layered on top of content, Docuvera embeds governance directly into how regulated data is created, managed, and reused.
By maintaining a centralized, governed data foundation, Docuvera ensures that rules are enforced at the data level across regulated content and data reuse workflows. Approval status, lineage, and reuse constraints travel with the data wherever it is used, ensuring that downstream applications inherit governance controls rather than re-implementing them through process. Teams no longer need to re-establish trust with each reuse; trust is inherent.
Importantly, Docuvera complements existing systems rather than replacing them. Regulatory, labeling, and operational platforms continue to perform their roles, but they draw from a common governed data source. This preserves validation integrity while eliminating fragmentation.
The impact on compliance outcomes
When governance is embedded upstream, compliance improves measurably. Inconsistencies decline because divergence is prevented. Audit readiness increases because lineage is explicit, traceable, and consistently maintained across the data lifecycle, aligning with regulatory expectations for attributable, legible, contemporaneous, original, and accurate data, alongside the additional enduring, available, complete, consistent, and credible expectations commonly grouped as ALCOA+, as outlined in the WHO’s guidance on good data and record management practices.
Compliance teams move from reactive oversight to proactive stewardship. Instead of policing downstream outputs, they define and maintain governance rules that scale with the organization.
The impact on speed and efficiency
At the same time, speed increases. Submission timelines shorten because revalidation cycles disappear. Updates propagate faster because changes are made once. Global execution accelerates because teams work in parallel from the same trusted data.
These gains are durable. They do not depend on heroic effort or exceptional individuals. They are structural outcomes of how data is governed.
Governance as a strategic capability
As regulatory requirements continue to evolve and portfolios become more complex, governance must scale in a way that remains demonstrable, auditable, and repeatable across markets.
Process-heavy models cannot keep up. Data-centric governance can.
By investing in centralized, governed data management—enabled by platforms like Docuvera—organizations transform governance from a necessary burden into a strategic capability. Compliance and speed cease to be tradeoffs. They become mutually reinforcing outcomes of sound data architecture.
Speed follows when governance is built into data
The fastest pharma organizations are not those that compromise on compliance. They are those that understand where governance belongs. When governance is embedded into data, not bolted on through process, both compliance and speed improve.
Centralized data governance is not an IT initiative. It is an operating model decision. Organizations that make this shift gain clarity, confidence, and momentum across the entire product lifecycle—without increasing risk.
FAQs | Centralized Pharma Data Governance
Sources