2026 Pharma Readiness: Regulatory Readiness Is Now an Enterprise Capability
Regulatory readiness has shifted from a supporting function to a defining enterprise capability in the pharmaceutical industry, and that is changing how a regulatory operating model pharma must be designed for regulatory readiness 2026. Modern regulatory execution is increasingly shaped by electronic submission expectations, structured content requirements, global data standards, and new AI governance obligations across major markets. Understanding the regulatory operating model pharma is crucial for maintaining compliance and enhancing operational effectiveness, as the regulatory operating model pharma directly impacts how pharmaceutical companies approach their regulatory processes.
As regulatory scrutiny intensifies and global requirements evolve, organizations are discovering that success is no longer determined by whether individual regulations are met. Instead, it hinges on how consistently, efficiently, and predictably regulatory work is executed across products, regions, and time.
Understanding the regulatory operating model pharma is crucial for maintaining compliance and enhancing operational effectiveness.
The regulatory operating model pharma must be adaptable to meet new challenges, ensuring that regulatory teams are prepared for the evolving landscape of compliance and operational efficiency.
Ultimately, the success of a regulatory operating model pharma will depend on its ability to scale with the changing regulatory environment, ensuring that organizations remain competitive and compliant.
By the end of 2026, regulatory readiness will be measured less by isolated compliance milestones and more by the strength, scalability, and resilience of the regulatory operating model itself. Health authorities are signaling this shift through increasing expectations for electronic submission structure and lifecycle management, structured master data, and more governed AI use in regulated settings. The FDA’s guidance on electronic submissions and eCTD, EMA’s SPOR/IDMP framework, and the EU AI Act all point in the same direction: content, data, and governance must be managed as connected operating-model capabilities, not isolated tasks.
Consequently, organizations must prioritize the optimization of their regulatory operating model pharma to achieve greater efficiency and compliance.
This fragility in the current regulatory operating model pharma can hinder the efficiency needed to navigate the complexities of modern regulations.
To combat these inefficiencies, companies must evolve their regulatory operating model pharma to embrace modern challenges and improve compliance.
The Hidden Fragility of Incremental Operating Models
Ultimately, the objective of any regulatory operating model pharma is to streamline processes and enhance regulatory efficiency across the board.
For many pharmaceutical organizations, regulatory operating models have evolved incrementally rather than intentionally. Processes are layered on top of existing ones. New tools are introduced to address immediate gaps. Institutional knowledge accumulates within individuals rather than being embedded in systems. The difference between incremental and intentional operating models can significantly impact regulatory efficiency, influencing the organization’s ability to adapt and succeed in a rapidly changing environment.

While these approaches may sustain day-to-day operations, they introduce structural fragility as complexity increases. Regulatory teams often spend disproportionate time coordinating work, reconciling inconsistencies, and validating information that should already be trusted. The result is slower submissions, reactive fire drills during inspections, and limited capacity to absorb regulatory change.
Importantly, these challenges are not caused by lack of expertise. They are symptoms of operating models that were never designed to scale under modern regulatory demands—particularly in environments where content reuse, global harmonization, and lifecycle traceability are becoming expectations rather than best practices. Ultimately, inefficient regulatory models can slow progress across development and delay access to medicines for patients.
Key Factors That Define a Modern Regulatory Operating Model
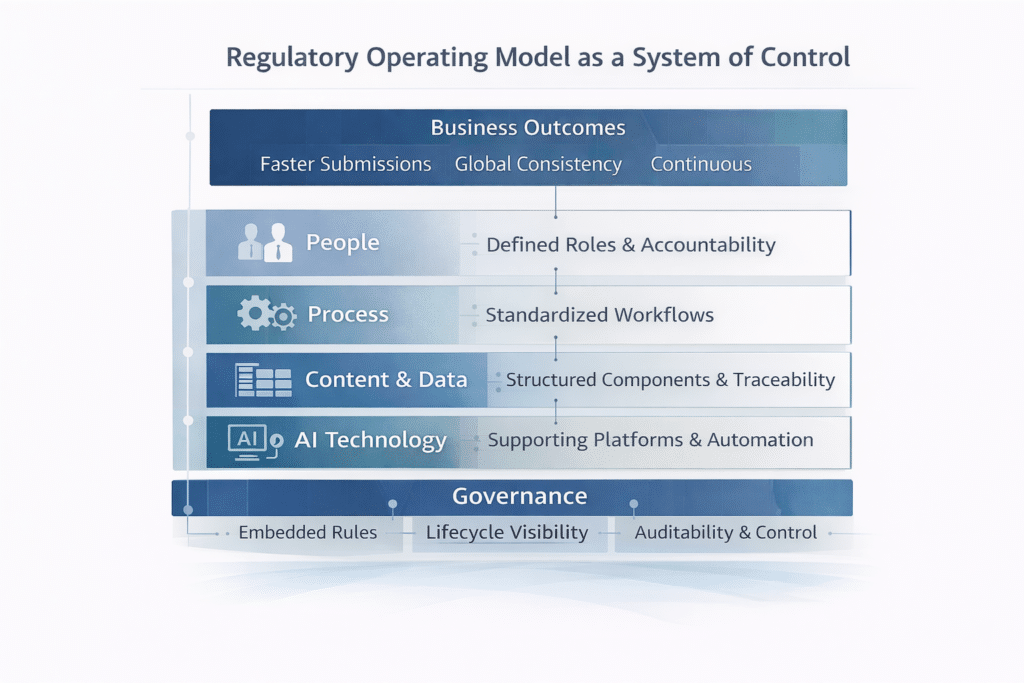
A modern regulatory operating model in pharma aligns people, processes, data, and technology around a shared objective: consistent, controlled, and efficient regulatory execution.
In practice, this means:
It is essential for organizations to focus on key therapeutic areas such as oncology, cardiometabolic, neurodegenerative, immunologic, and obesity/metabolic conditions, as these are central to innovation and market growth. Essential requirements for a modern regulatory operating model also include robust compliance, advanced data management, and agile adaptation to evolving regulations.
The role of digital health, genomics, and AI-driven innovation is increasingly important in regulatory operations, shaping how companies approach submissions and compliance. Industry leaders are guiding these strategic initiatives, investing in new platforms and driving digital transformation across the sector. Pharmaceutical companies must prioritize upskilling to address workforce gaps in AI literacy and data analytics to remain competitive. McKinsey notes that generative AI is already reshaping pharma operations, including accelerated drug discovery, more efficient trials, and faster regulatory approvals.
Organizations are increasingly integrating agentic AI to autonomously perform complex tasks, further improving efficiency in regulatory programs. As this evolves, the operating model matters even more: the more automated the workflow, the more important governance, traceability, and decision rights become. Agentic AI is expected to drive new performance gains in biopharma development when paired with strong operating models.
Docuvera operationalizes these principles by embedding governance, reuse rules, and lifecycle visibility directly into regulatory workflows—making the operating model executable, not aspirational. Explore how this works in practice on the Docuvera platform.
Moving Beyond Document-Centric Regulatory Thinking with Digital Innovation
One of the most significant shifts in modern regulatory operations is the move away from document-centric thinking. Traditional regulatory models rely on static documents that are copied, edited, and redistributed across teams and geographies. Over time, this leads to duplicated effort, version conflicts, and uncertainty about which information is authoritative.
In contrast, modern operating models emphasize structured content and reusable components for regulatory submissions, aligning with FDA electronic submission standards and EMA IDMP/SPOR data models that prioritize interoperability and consistency.
Structured content also enables more reliable and consistent use of clinical and regulatory data across submissions, supporting efficient development and approval workflows for targeted therapies and new modalities. Changes are made at the source and propagated systematically, rather than re-implemented manually in dozens of downstream documents.
This shift is foundational—not incremental. It enables traceability, lifecycle management, and responsiveness that document-based approaches cannot deliver at scale. Docuvera supports this transition by replacing document assembly with governed, component-based regulatory execution.
As regulatory operations become more interconnected and AI-driven, ensuring data integrity and robust cybersecurity protocols is critical to protect sensitive information and maintain compliance amid rising cyber threats. The FDA emphasizes this need through its cybersecurity guidance for regulated digital systems.
Strategically, the regulatory operating model pharma must support innovation while ensuring compliance with evolving regulations.
Organizations that prioritize their regulatory operating model pharma will be better equipped to navigate regulatory changes and maintain compliance.
Designing for 2026: Intentional Operating Model Choices in Drug Development
Building a regulatory operating model in pharma fit for regulatory readiness 2026 requires intentional design decisions. In today’s world, regulatory readiness is a global challenge, requiring international coordination and alignment across markets. Pharmaceutical readiness for 2026 also involves the strategic integration of AI, automation, resilient supply chains, and a focus on personalized medicine.
Centralization plays a critical role, particularly for content governance, standards, and data management. At the same time, effective models support distributed execution, enabling global and cross-functional teams to work in parallel within clearly defined boundaries.
Key design principles include:
Organizations that make these choices deliberately are better positioned to absorb regulatory change without disruption.
Technology’s Role: Enabling, Not Leading
Technology is a critical enabler of modern regulatory operating models—but it is not the starting point. Successful organizations first define how regulatory work should flow, how decisions should be made, and how content should be governed across its lifecycle. Industry leaders emphasize that AI delivers value only when paired with strong operating models.
In 2026, the industry will see a significant rise in hybrid roles such as digital medical advisors and AI-clinical analysts, reflecting the shift as digital health roles become more mainstream and reshape workforce needs and operating models. Technology then reinforces these decisions by embedding controls, automating workflows, enabling reuse, and providing real-time visibility. When introduced without this foundation, technology risks digitizing inefficiency rather than eliminating it.
As we look toward 2026 pharma readiness, AI is moving from experimental to foundational infrastructure, streamlining R&D, manufacturing, and commercial operations. AI is already transforming drug development workflows across the value chain.
How Docuvera Enables a Modern Regulatory Operating Model
Docuvera supports modern regulatory operating models by providing a single, governed platform for managing regulatory content across its entire lifecycle. It centralizes content while enabling controlled collaboration across functions and regions—reducing fragmentation without constraining execution. Learn more about Docuvera’s approach on the platform overview and regulatory operations solution.
Key capabilities include:
In summary, an efficient regulatory operating model pharma is essential for maintaining compliance and ensuring success in the rapidly changing pharmaceutical landscape.
Docuvera enables regulatory submissions for advanced therapies, including cell and gene therapies, antibody-drug conjugates, and oncology products, supporting tailored commercial strategies and compliance with evolving regulatory requirements.
By shifting regulatory work from document assembly to governed content management, Docuvera helps organizations respond to regulatory change efficiently—without rework or loss of control.
From Cost Center to Competitive Advantage
As regulatory expectations continue to evolve, organizations that invest in scalable operating models will be better positioned to adapt. In parallel, regulatory and supply chain integrity requirements continue to expand, particularly in the United States, where DSCSA requirements are driving more interoperable and traceable systems.
By 2026, regulatory readiness will increasingly distinguish organizations that react to change from those that lead through it. The FDA’s electronic submission framework, the EU AI Act, and EMA’s SPOR/IDMP standards all reinforce the same strategic direction.
A modern regulatory operating model does more than reduce risk. It improves speed, consistency, and confidence across the regulatory lifecycle. In doing so, it transforms compliance from a cost of doing business into a sustainable competitive advantage.