Billions are moving. The documentation problem that follows every closed deal is not on the term sheet.
In February, Gilead Sciences agreed to acquire Arcellx for an implied equity value of $7.8 billion, securing full control of anitocabtagene autoleucel (anito-cel), a late-stage CAR-T therapy for multiple myeloma. On March 6, 2026, Servier announced a $2.5 billion acquisition of Day One Biopharmaceuticals and its approved pediatric low-grade glioma therapy, Ojemda. On March 23, 2026, Gilead announced a second deal, acquiring Ouro Medicines for up to $2.18 billion to add OM336 (gamgertamig), a clinical-stage BCMAxCD3 T-cell engager for autoimmune diseases. And on March 25, 2026, Merck announced a $6.7 billion acquisition of Terns Pharmaceuticals, adding TERN-701, an oral, investigational allosteric BCR-ABL inhibitor for chronic myeloid leukemia.
Four major deals. More than $19 billion in announced aggregate value. And in each announcement, the emphasis is on pipeline, portfolio, or strategic rationale rather than on what regulatory teams will face the morning after the deal closes.
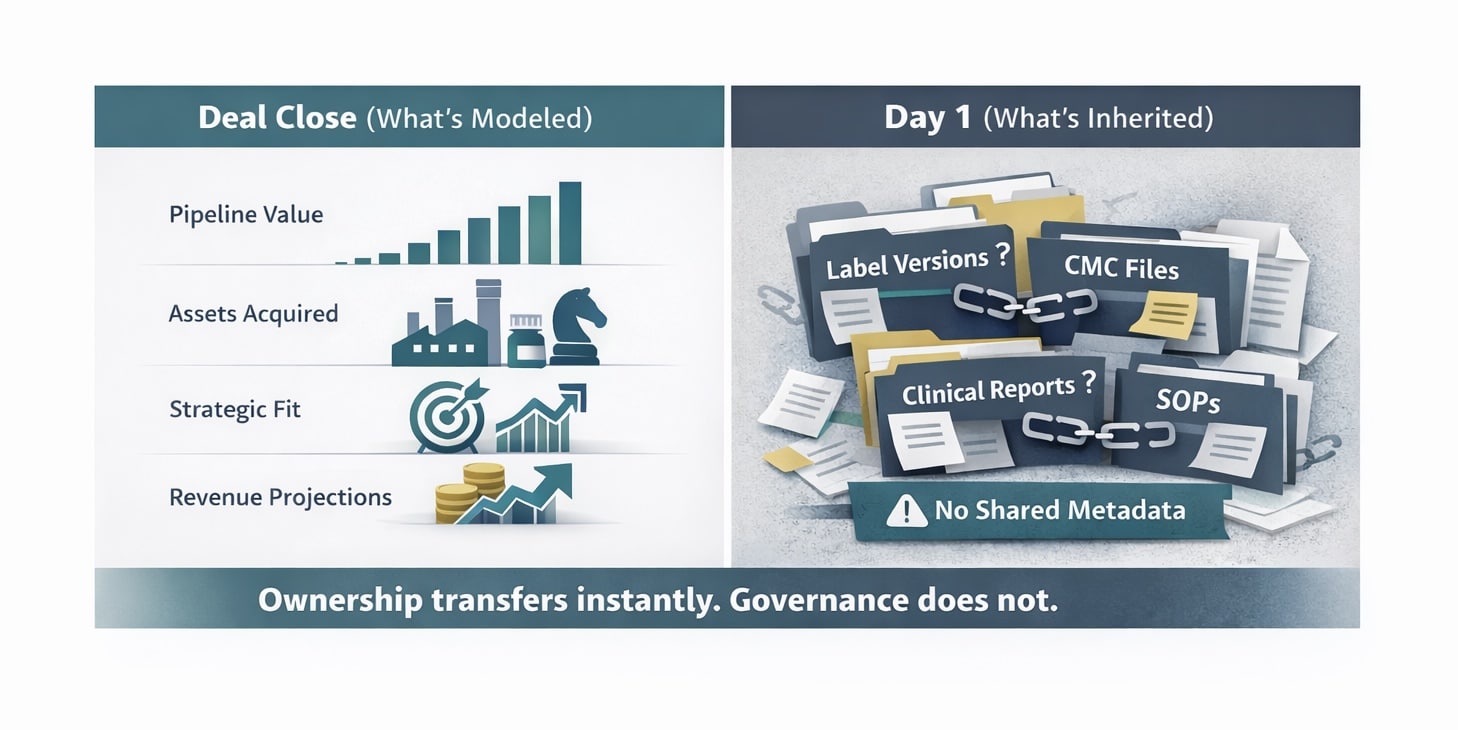
The Pharma M&A Integration Problem That Starts at Close
Every acquisition in regulated pharma triggers the same sequence. Two organizations with separate content environments must produce a single, audit-ready regulatory record. The acquiring organization inherits product labels, CMC documentation, clinical study reports, safety documentation, SOPs, and submission histories – all governed under different systems, different approval workflows, and different metadata standards, assuming metadata standards exist at all. In practice, those records must still support traceability, audit trail integrity, and electronic-record controls under frameworks such as 21 CFR Part 11 and EU GMP Annex 11.
This is not a data migration problem. It is a regulatory continuity problem. And it begins the moment ownership transfers.
A labeling team inheriting an acquired portfolio has an immediate inventory challenge. Labels may live in multiple repositories with no shared naming convention. Version histories may follow different numbering schemes, or no formal scheme. Approval chains documented under the acquired company’s quality framework may not translate into the acquirer’s. Every gap in version lineage, every undefined content component, every missing review record extends the period before the acquired portfolio is defensible under a single governance model and ready to support future eCTD lifecycle activity under a unified submission history.
The same challenge runs through CMC documentation, where process descriptions and specifications must reconcile against the acquirer’s quality systems. Through clinical documentation, where study reports and regulatory correspondence must integrate into a unified submission history. Through SOPs, where procedural differences must be identified, assessed, and governed under a single quality management structure.
Why Pharma M&A Due Diligence Doesn’t Catch It
Bain’s 2026 pharma M&A analysis characterizes the current cycle as a shift from pipeline chasing to platform and production control. Strategic pharma deal value rose 79% through November 15, 2025, with the average deal size climbing by more than 80%. Bain’s broader 2026 M&A outlook found that 80% of M&A executives expect to sustain or increase deal activity in 2026. What those reports do not address is what happens to regulatory documentation when two organizations with unequal content maturity merge their portfolios.
The omission is structural, not accidental. Content operations infrastructure often appears in deal models as headcount, tooling spend, and document management licensing costs. It reads as IT overhead. Deal teams are typically organized around the variables that most visibly drive valuation; documentation architecture usually becomes more visible post-close, when the actual integration timeline becomes clear. That matters because regulatory and compliance due diligence is already recognized as a core workstream in FDA-regulated transactions
There is no widely adopted standard for assessing documentation readiness at the point of due diligence. There is no common content maturity scorecard used across pharma transactions. There is no way to determine, from outside, how long it will take to bring an acquired labeling portfolio into the acquirer’s governance framework. The answer depends on questions that rarely appear in the data room: How is content stored? Is version history traceable? Are document components discrete or embedded in monolithic files? Does the organization have defined metadata standards for regulatory content?
The acquirer discovers the answers post-close.
The Structural Variable
Content architecture is an integration variable with direct implications for post-acquisition timeline and regulatory risk.
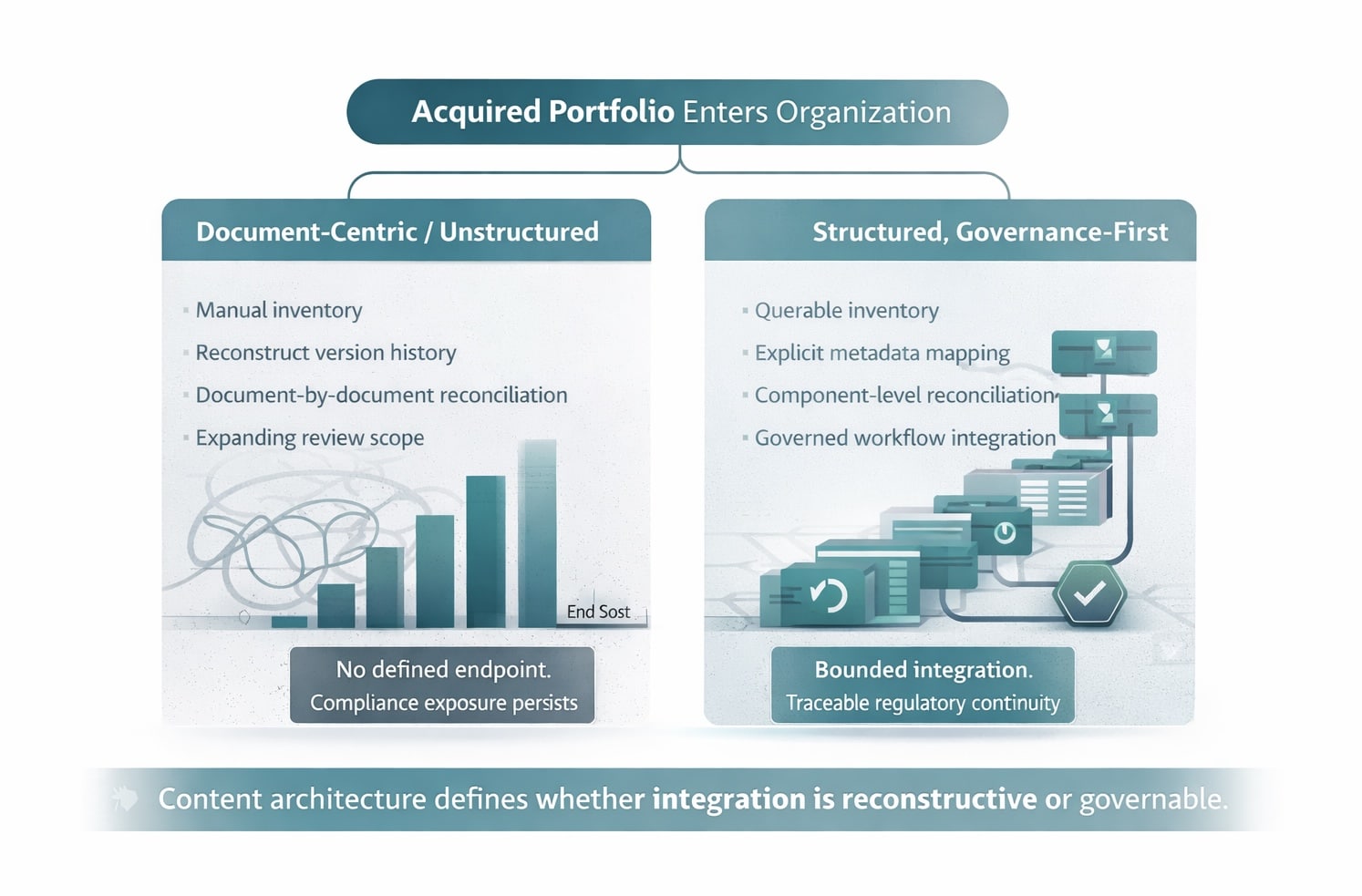
Organizations managing regulatory content in structured, component-based environments bring a defined integration surface to the transaction. Version history is queryable. Metadata standards are explicit and comparable. Content components can be mapped, reconciled, and migrated into a unified governance framework in a planned sequence. The integration work is more bounded and more inspectable.
Organizations managing content in document-centric, siloed environments bring opaque archives. There is no clean inventory because the inventory process itself is the first task. Version traceability must be reconstructed rather than retrieved. Decisions that a governed system makes automatically – which version is current, which components are shared across products, which markets have received which label versions – must be made manually, one document at a time.
That difference, compounded across labeling, CMC, clinical, and quality documentation, can materially change the post-close harmonization timeline. The regulatory requirements that govern this content – 21 CFR Part 11, EU GMP Annex 11, and the broader submission and dossier expectations reflected in eCTD and CTD structures – do not pause for deal activity. The acquirer assumes responsibility for the acquired portfolio at close.
What Structured Content Readiness Enables in Pharma M&A Integration
Docuvera’s governance-first structured content authoring platform authors content as discrete, governed components with defined metadata, traceable version control at the element level, and governed workflow at each review and approval stage. That architecture creates a defined integration surface for acquisitions.
When a new product portfolio enters scope, the path is explicit: map acquired content to the established governance framework, establish version lineage for each document artifact, and bring the full portfolio under a single content governance model. The components are discrete. The standards are explicit. The scope of integration work is more knowable before the work begins.
Organizations working from unstructured document environments face the alternative. A manual inventory followed by artifact-by-artifact assessment, reformatting, and reconciliation. No bounded scope. No defined endpoint until the last document is resolved.
For regulatory affairs, labeling, and CMC leaders at organizations operating in an active M&A environment, this distinction is not abstract. The content environment that exists at the point a deal closes determines how long the compliance exposure persists. An independent analysis of structured content authoring approaches in life sciences provides additional context on the architectural differences between platform categories.
The Calculus for 2026
Pharma M&A is not slowing. Bain’s executive survey found that 80% of M&A executives expect activity to sustain or increase in 2026. For any large pharma organization, the probability of involvement in a transaction this year – as buyer, target, or both – is not low.
The organizations that have invested in governed, structured content infrastructure carry a measurable advantage. Lower integration cost. A shorter path to regulatory continuity. A defined compliance posture during the transition period. The organizations that have not will encounter the difference in timeline, in cost, and in the compliance exposure that remains open until harmonization is complete.
Content architecture is preparation for any event that requires two regulated content environments to become one.
Frequently Asked Questions
Editorial note: This analysis is based on public company announcements, regulatory guidance, and industry reports available as of April 2026. It is intended as strategic analysis of post-acquisition regulatory content governance, not legal advice.
Sources
- Gilead Sciences. “Gilead Sciences to Acquire Arcellx to Maximize Long-Term Potential of Anito-cel.” Feb. 23, 2026. gilead.com
- Servier. “Servier and Day One Biopharmaceuticals Announce Acquisition to Expand Servier’s Rare Oncology Portfolio.” March 6, 2026. servier.com
- Bloomberg. “Gilead Nears Deal to Buy Ouro Medicines for Its Autoimmune Drugs.” March 23, 2026. bloomberg.com
- Merck. “Merck to Acquire Terns Pharmaceuticals, Inc., Expanding Its Hematology Pipeline With TERN-701.” March 25, 2026. merck.com
- Bain & Company. “M&A in Pharmaceuticals: Bigger, Bolder, and Far More Strategic.” 2026 M&A Report. bain.com
- Bain & Company. “Looking Ahead to 2026: Getting a Boost from the Great Rebound.” 2026 M&A Report. bain.com