On March 5, 2026, FDA published a final rule establishing a uniform 12-digit National Drug Code (NDC) format. The rule takes effect on March 7, 2033. An enforcement flexibility window extends to March 2036, coinciding with the implementation of the FDA 12-digit NDC Rule.
Seven years sounds like a long runway. For large pharmaceutical portfolios, it is not.
The compliance question here is not technical. It is operational: How does a labeling team govern the controlled update of every affected label, barcode, and regulatory document across products, markets, and formats — without turning a format change into a multi-year remediation campaign?
Understanding the nuances of the FDA 12-digit NDC Rule is essential for compliance.
All affected products must align with the FDA 12-digit NDC Rule to avoid compliance issues.
The FDA 12-digit NDC Rule necessitates a shift in how products are coded and identified.
Most labeling teams have not started asking that question yet.
What the Rule Actually Requires
The new format adopts a 6-4-2 segmentation structure: a six-digit labeler code, a four-digit product code, and a two-digit package code. Existing 10-digit NDCs convert by adding leading zeros to each segment. No labeler code reissuance is required. The structural change propagates, however, across every drug label currently carrying a 10-digit code.
The barcode implications compound the scope of work. UPC-A barcodes cannot encode a 12-digit NDC. FDA’s final rule accepts GS1-128, GS1 DataBar, and 2D DataMatrix as compliant symbologies. GS1 has developed a new Application Identifier (AI 715) specifically to support 12-digit encoding across these formats. Organizations using UPC-A today face a symbology migration on top of the NDC format to change itself.
Adapting to the FDA 12-digit NDC Rule requires a comprehensive review of all labeling practices.
There is a second layer worth noting early. For organizations also subject to Drug Supply Chain Security Act (DSCSA) serialization requirements, the transition creates a consolidation opportunity. A single 2D DataMatrix barcode encoded with AI 715 can carry the 12-digit NDC alongside serial number, lot number, and expiration date. Beginning in 2033, one barcode can satisfy both NDC labeling and DSCSA product identifier requirements in a single scan. That alignment is operationally significant, but only for organizations that plan it deliberately.
Why Seven Years Is Not What It Appears
Consider the actual scope of work for a Top 50 pharma organization: hundreds of marketed products, multiple geographies, multiple regulatory jurisdictions. Each product label requires a controlled change — updated NDC, updated barcode symbology, updated artwork, updated regulatory submissions. Each of those changes triggers a documentation lifecycle event. Each lifecycle event must be traceable, version-controlled, and audit-ready.
Understanding the requirements of the FDA 12-digit NDC Rule is crucial for compliance.
The volume alone creates real scheduling pressure. An organization with 200 or more marketed products cannot run sequential label updates in a compressed final window. Regulatory operations, quality review, and artwork production would face simultaneous demand that most teams cannot absorb. Staggered, planned migration over several years is the only approach that works at portfolio scale. That planning does not start in 2030.
In the context of the FDA 12-digit NDC Rule, compliance will require careful planning and execution.
Organizations must prioritize the FDA 12-digit NDC Rule in their operational strategy moving forward.
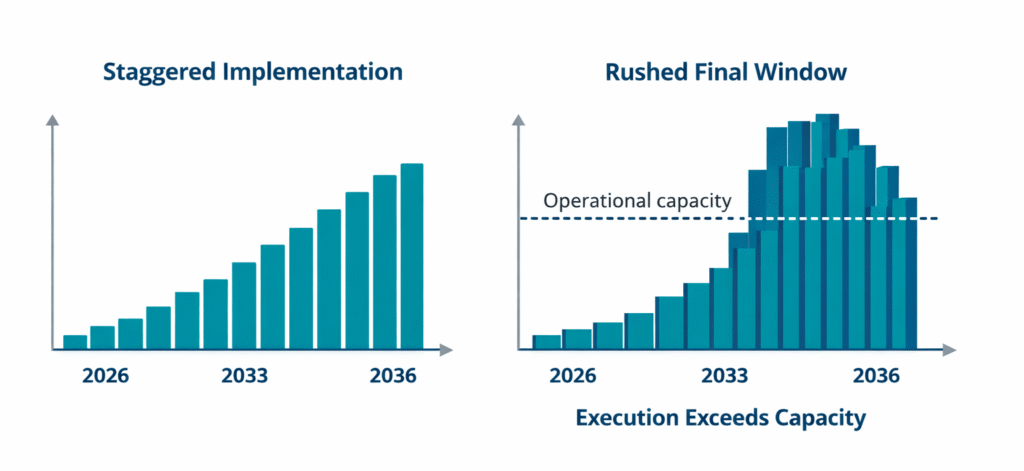
The implications of the FDA 12-digit NDC Rule will ripple through many aspects of pharmaceutical operations.
This is not a technology argument. It is a resource and governance argument.
Now layer in the operational reality most labeling teams already navigate. NDC data is maintained in spreadsheets. Artwork files living outside controlled systems. Barcode specifications tracked informally across functions. In that environment, every label touched creates reconciliation risk. Every manual handoff introduces error potential. Every uncontrolled version creates audit exposure.
Faegre Drinker’s published analysis of the final rule notes that the compliance burden will correlate with portfolio size. Organizations with fragmented labeling workflows face proportionally greater operational exposure.
Companies must align their practices with the FDA 12-digit NDC Rule to maintain compliance.
The FDA 12-digit NDC Rule is a critical component of future drug labeling strategies.
This Is a Content Governance Problem, Not a Barcode Project
The framing matters.
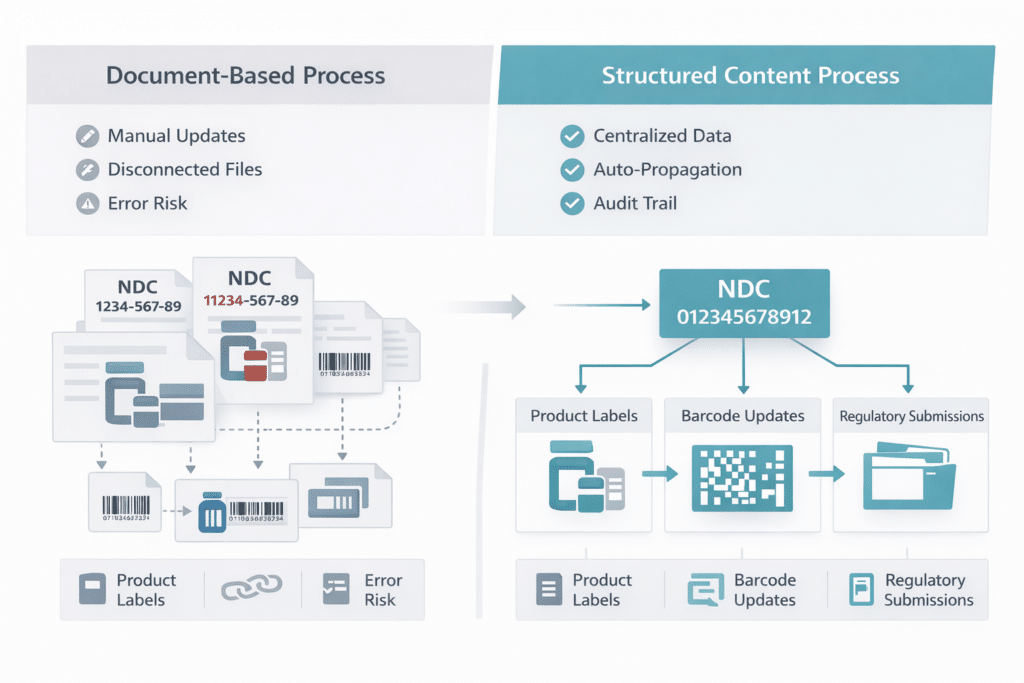
The label is a regulated document. The NDC is a data element within that document. The barcode is a rendered output of that data element. When any part of this chain changes, the entire chain must update in a controlled, auditable sequence. Static document environments cannot manage that reliably across large portfolios.
Effective structured content authoring is key to managing the FDA 12-digit NDC Rule changes.
This is the operational case for structured content authoring as regulatory infrastructure — not as a content management preference, but as the mechanism that makes portfolio-wide label changes executable without manual remediation at every step.
In a governed structured environment, an NDC format change is a controlled data update. It propagates through every affected artifact in a traceable sequence. In a disconnected document environment, the same change is a manual project, touching each product label individually. At 200-plus products, the difference between those two scenarios is the difference between a managed compliance cycle and a resource crisis.
Regulatory operations teams have already run at capacity. Adding a portfolio-wide label change without structural support creates resource contention, delays submissions, and elevates compliance risk.
The New Risk Surface in the Pharma Content Lifecycle addresses the broader pattern: when content is ungoverned at the source, every downstream compliance event costs disproportionately more to execute.
The FDA 12-digit NDC Rule represents a significant shift in how labeling will be managed across the industry.
Docuvera’s governance-first labeling content platform addresses this directly. Content components are version-controlled at the element level. Metadata is governed at authoring, not appended downstream. Label lifecycle events are traceable from source through output. When a data element like the NDC changes, the update flows through every dependent artifact in a controlled sequence. For organizations managing large product portfolios, that is not an enhancement. It is the operational prerequisite.
The Planning Window Is Open
FDA gave the industry seven years. That is a planning window, not a deferral. It closes faster than any portfolio projection makes it appear.
The first step is an honest operational assessment. Can your labeling content environment propagate a format change across your full product portfolio in a controlled, traceable, audit-ready sequence? If that answer requires qualification, the work has already begun — whether your team has started or not.
Get in touch with our team to learn more.
Frequently Asked Questions: 12-Digit NDC Planning
Understanding the FDA 12-digit NDC Rule
Preparation for the FDA 12-digit NDC Rule will involve significant adjustments in labeling protocols.
Sources